Polysialic acid is required for dopamine D2 receptor-mediated plasticity involving inhibitory circuits of the rat medial prefrontal cortex
- PMID: 22216301
- PMCID: PMC3247286
- DOI: 10.1371/journal.pone.0029516
Polysialic acid is required for dopamine D2 receptor-mediated plasticity involving inhibitory circuits of the rat medial prefrontal cortex
Abstract
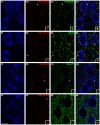
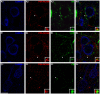
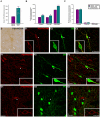
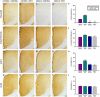
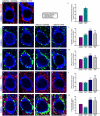
Decreased expression of dopamine D2 receptors (D2R), dysfunction of inhibitory neurotransmission and impairments in the structure and connectivity of neurons in the medial prefrontal cortex (mPFC) are involved in the pathogenesis of schizophrenia and major depression, but the relationship between these changes remains unclear. The polysialylated form of the neural cell adhesion molecule (PSA-NCAM), a plasticity-related molecule, may serve as a link. This molecule is expressed in cortical interneurons and dopamine, via D2R, modulates its expression in parallel to that of proteins related to synapses and inhibitory neurotransmission, suggesting that D2R-targeted antipsychotics/antidepressants may act by affecting the plasticity of mPFC inhibitory circuits. To understand the role of PSA-NCAM in this plasticity, rats were chronically treated with a D2R agonist (PPHT) after cortical PSA depletion. PPHT-induced increases in GAD67 and synaptophysin (SYN) neuropil expression were blocked when PSA was previously removed, indicating a role for PSA-NCAM in this plasticity. The number of PSA-NCAM expressing interneuron somata also increased after PPHT treatment, but the percentages of these cells belonging to different interneuronal subpopulations did not change. Cortical pyramidal neurons did not express PSA-NCAM, but puncta co-expressing this molecule and parvalbumin could be found surrounding their somata. PPHT treatment increased the number of PSA-NCAM and parvalbumin expressing perisomatic puncta, but decreased the percentage of parvalbumin puncta that co-expressed SYN. PSA depletion did not block these effects on the perisomatic region, but increased further the number of parvalbumin expressing puncta and increased the percentage of puncta co-expressing SYN and parvalbumin, suggesting that the polysialylation of NCAM may regulate perisomatic inhibition of mPFC principal neurons. Summarizing, the present results indicate that dopamine acting on D2R influences structural plasticity of mPFC interneurons and point to PSA-NCAM as a key player in this remodeling.
© 2011 Castillo-Gómez et al.
Conflict of interest statement
Figures
References
-
- Castren E. Is mood chemistry? Nat Rev Neurosci. 2005;6:241–246. - PubMed
-
- Lewis DA, Gonzalez-Burgos G. Neuroplasticity of neocortical circuits in schizophrenia. Neuropsychopharmacology. 2008;33:141–165. - PubMed
-
- Benes FM, Berretta S. GABAergic interneurons: implications for understanding schizophrenia and bipolar disorder. Neuropsychopharmacology. 2001;25:1–27. - PubMed
-
- Daskalakis ZJ, Fitzgerald PB, Christensen BK. The role of cortical inhibition in the pathophysiology and treatment of schizophrenia. Brain Res Rev. 2007;56:427–442. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

