Novel role for non-muscle myosin light chain kinase (MLCK) in hyperoxia-induced recruitment of cytoskeletal proteins, NADPH oxidase activation, and reactive oxygen species generation in lung endothelium
- PMID: 22219181
- PMCID: PMC3308820
- DOI: 10.1074/jbc.M111.294546
Novel role for non-muscle myosin light chain kinase (MLCK) in hyperoxia-induced recruitment of cytoskeletal proteins, NADPH oxidase activation, and reactive oxygen species generation in lung endothelium
Abstract
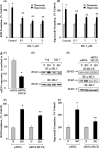
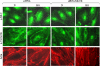
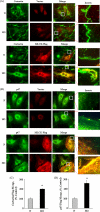
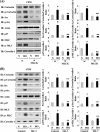
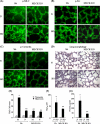
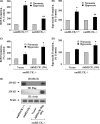
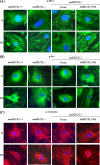
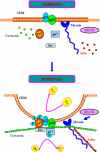
We recently demonstrated that hyperoxia (HO) activates lung endothelial cell NADPH oxidase and generates reactive oxygen species (ROS)/superoxide via Src-dependent tyrosine phosphorylation of p47(phox) and cortactin. Here, we demonstrate that the non-muscle ~214-kDa myosin light chain (MLC) kinase (nmMLCK) modulates the interaction between cortactin and p47(phox) that plays a role in the assembly and activation of endothelial NADPH oxidase. Overexpression of FLAG-tagged wild type MLCK in human pulmonary artery endothelial cells enhanced interaction and co-localization between cortactin and p47(phox) at the cell periphery and ROS production, whereas abrogation of MLCK using specific siRNA significantly inhibited the above. Furthermore, HO stimulated phosphorylation of MLC and recruitment of phosphorylated and non-phosphorylated cortactin, MLC, Src, and p47(phox) to caveolin-enriched microdomains (CEM), whereas silencing nmMLCK with siRNA blocked recruitment of these components to CEM and ROS generation. Exposure of nmMLCK(-/-) null mice to HO (72 h) reduced ROS production, lung inflammation, and pulmonary leak compared with control mice. These results suggest a novel role for nmMLCK in hyperoxia-induced recruitment of cytoskeletal proteins and NADPH oxidase components to CEM, ROS production, and lung injury.
Figures



Similar articles
-
Dynamin 2 and c-Abl are novel regulators of hyperoxia-mediated NADPH oxidase activation and reactive oxygen species production in caveolin-enriched microdomains of the endothelium.J Biol Chem. 2009 Dec 11;284(50):34964-75. doi: 10.1074/jbc.M109.013771. Epub 2009 Oct 15. J Biol Chem. 2009. PMID: 19833721 Free PMC article.
-
Regulation of hyperoxia-induced NADPH oxidase activation in human lung endothelial cells by the actin cytoskeleton and cortactin.J Biol Chem. 2007 Aug 10;282(32):23284-95. doi: 10.1074/jbc.M700535200. Epub 2007 Jun 11. J Biol Chem. 2007. PMID: 17562703
-
Phospholipase D-mediated activation of IQGAP1 through Rac1 regulates hyperoxia-induced p47phox translocation and reactive oxygen species generation in lung endothelial cells.J Biol Chem. 2009 May 29;284(22):15339-52. doi: 10.1074/jbc.M109.005439. Epub 2009 Apr 14. J Biol Chem. 2009. PMID: 19366706 Free PMC article.
-
Regulation of NADPH oxidase in vascular endothelium: the role of phospholipases, protein kinases, and cytoskeletal proteins.Antioxid Redox Signal. 2009 Apr;11(4):841-60. doi: 10.1089/ars.2008.2231. Antioxid Redox Signal. 2009. PMID: 18828698 Free PMC article. Review.
-
Pulmonary endothelial cell NOX.Am J Respir Cell Mol Biol. 2012 Aug;47(2):129-39. doi: 10.1165/rcmb.2010-0331RT. Epub 2012 Apr 12. Am J Respir Cell Mol Biol. 2012. PMID: 22499852 Free PMC article. Review.
Cited by
-
Inhibition of myosin light chain kinase reduces NADPH oxidase-mediated oxidative injury in rat brain following cerebral ischemia/reperfusion.Naunyn Schmiedebergs Arch Pharmacol. 2015 Sep;388(9):953-63. doi: 10.1007/s00210-015-1125-2. Epub 2015 Apr 29. Naunyn Schmiedebergs Arch Pharmacol. 2015. PMID: 25920934
-
Nrf2 Activation Protects Against Organic Dust and Hydrogen Sulfide Exposure Induced Epithelial Barrier Loss and K. pneumoniae Invasion.Front Cell Infect Microbiol. 2022 Apr 19;12:848773. doi: 10.3389/fcimb.2022.848773. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35521223 Free PMC article.
-
MicroRNA and mRNA analysis of angiotensin II-induced renal artery endothelial cell dysfunction.Exp Ther Med. 2020 Jun;19(6):3723-3737. doi: 10.3892/etm.2020.8613. Epub 2020 Mar 19. Exp Ther Med. 2020. PMID: 32346437 Free PMC article.
-
Human and mouse microarrays-guided expression analysis of membrane protein trafficking-related genes in MDCK cells, a canine epithelial model for apical and basolateral differential protein targeting.Biochim Open. 2017 Apr 30;4:119-126. doi: 10.1016/j.biopen.2017.04.002. eCollection 2017 Jun. Biochim Open. 2017. PMID: 29450149 Free PMC article.
-
Neuropeptide Y: An Update on the Mechanism Underlying Chronic Intermittent Hypoxia-Induced Endothelial Dysfunction.Front Physiol. 2021 Aug 27;12:712281. doi: 10.3389/fphys.2021.712281. eCollection 2021. Front Physiol. 2021. PMID: 34512386 Free PMC article. Review.
References
-
- Li J. M., Shah A. M. (2002) Intracellular localization and preassembly of the NADPH oxidase complex in cultured endothelial cells. J. Biol. Chem. 277, 19952–19960 - PubMed
-
- Touyz R. M., Yao G., Quinn M. T., Pagano P. J., Schiffrin E. L. (2005) p47phox associates with the cytoskeleton through cortactin in human vascular smooth muscle cells. Role in NAD(P)H oxidase regulation by angiotensin II. Arterioscler. Thromb. Vasc. Biol. 25, 512–518 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

