Thiazolidinedione-dependent activation of sphingosine kinase 1 causes an anti-fibrotic effect in renal mesangial cells
- PMID: 22221312
- PMCID: PMC3417426
- DOI: 10.1111/j.1476-5381.2012.01824.x
Thiazolidinedione-dependent activation of sphingosine kinase 1 causes an anti-fibrotic effect in renal mesangial cells
Abstract
Background and purpose: PPARγ agonists [thiazolidinediones (TZDs)] are known to exert anti-fibrotic effects in the kidney. In addition, we previously demonstrated that sphingosine kinase 1 (SK-1) and intracellular sphingosine-1-phosphate (S1P), by reducing the expression of connective tissue growth factor (CTGF), have a protective role in the fibrotic process.
Experimental approach: Here, we investigated the effect of TZDs on intracellular sphingolipid levels and the transcriptional regulation of SK-1 in mesangial cells to evaluate potential novel aspects of the anti-fibrotic capacity of TZDs.
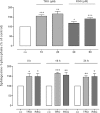
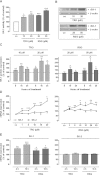
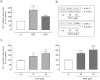
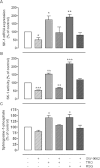
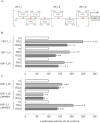
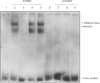
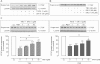
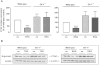
Key results: Stimulation with the TZDs, troglitazone and rosiglitazone, led to increased S1P levels in rat mesangial cells. This was paralleled by increased SK-1 activity as a consequence of direct effects of the TZDs on SK-1 expression. GW-9662, a PPARγ antagonist, inhibited the stimulating effect of TZDs on SK-1 mRNA and activity levels and intracellular S1P concentrations. Furthermore, SK-1 up-regulation by TZDs was functionally coupled with lower amounts of pro-fibrotic CTGF. SK-1 inhibition with SKI II almost completely abolished this effect in a dose-dependent manner. Moreover, the CTGF lowering effect of TZDs was fully blocked in MC isolated from SK-1 deficient mice (SK-1(-/-) ) as well as in glomeruli of SK-1(-/-) mice compared with wild-type mice treated with TRO and RSG.
Conclusion and implications: These data show that TZD-induced SK-1 up-regulation results in lower amounts of CTGF, demonstrating novel facets for the anti-fibrotic effects of this class of drugs.
© 2012 The Authors. British Journal of Pharmacology © 2012 The British Pharmacological Society.
Figures



Similar articles
-
PPARγ agonists upregulate sphingosine 1-phosphate (S1P) receptor 1 expression, which in turn reduces S1P-induced [Ca(2+)]i increases in renal mesangial cells.Biochim Biophys Acta. 2013 Nov;1831(11):1634-43. doi: 10.1016/j.bbalip.2013.07.011. Epub 2013 Jul 30. Biochim Biophys Acta. 2013. PMID: 23906789
-
Low-density lipoprotein induced expression of connective tissue growth factor via transactivation of sphingosine 1-phosphate receptors in mesangial cells.Mol Endocrinol. 2012 May;26(5):833-45. doi: 10.1210/me.2011-1261. Epub 2012 Mar 15. Mol Endocrinol. 2012. PMID: 22422617 Free PMC article.
-
Transforming growth factor-beta2 upregulates sphingosine kinase-1 activity, which in turn attenuates the fibrotic response to TGF-beta2 by impeding CTGF expression.Kidney Int. 2009 Oct;76(8):857-67. doi: 10.1038/ki.2009.297. Epub 2009 Aug 5. Kidney Int. 2009. PMID: 19657322
-
Targeting the sphingosine kinase/sphingosine 1-phosphate pathway to treat chronic inflammatory kidney diseases.Basic Clin Pharmacol Toxicol. 2014 Jan;114(1):44-9. doi: 10.1111/bcpt.12103. Epub 2013 Jul 15. Basic Clin Pharmacol Toxicol. 2014. PMID: 23789924 Review.
-
Sphingosine kinase, sphingosine-1-phosphate, and apoptosis.Biochim Biophys Acta. 2002 Dec 30;1585(2-3):193-201. doi: 10.1016/s1388-1981(02)00341-4. Biochim Biophys Acta. 2002. PMID: 12531554 Review.
Cited by
-
Effect of FTY720P on lipid accumulation in HEPG2 cells.Sci Rep. 2023 Nov 12;13(1):19716. doi: 10.1038/s41598-023-46011-4. Sci Rep. 2023. PMID: 37953311 Free PMC article.
-
Therapeutic Implications of Ferroptosis in Renal Fibrosis.Front Mol Biosci. 2022 May 17;9:890766. doi: 10.3389/fmolb.2022.890766. eCollection 2022. Front Mol Biosci. 2022. PMID: 35655759 Free PMC article. Review.
-
Sphingosine Kinase 2 Deficiency Attenuates Kidney Fibrosis via IFN-γ.J Am Soc Nephrol. 2017 Apr;28(4):1145-1161. doi: 10.1681/ASN.2016030306. Epub 2016 Oct 31. J Am Soc Nephrol. 2017. PMID: 27799486 Free PMC article.
-
Resveratrol Inhibits Lipopolysaccharide-Induced Extracellular Matrix Accumulation and Inflammation in Rat Glomerular Mesangial Cells by SphK1/S1P2/NF-κB Pathway.Diabetes Metab Syndr Obes. 2020 Nov 20;13:4495-4505. doi: 10.2147/DMSO.S278267. eCollection 2020. Diabetes Metab Syndr Obes. 2020. PMID: 33262625 Free PMC article.
-
Normalisation of surfactant protein -A and -B expression in the lungs of low birth weight lambs by 21 days old.PLoS One. 2017 Sep 26;12(9):e0181185. doi: 10.1371/journal.pone.0181185. eCollection 2017. PLoS One. 2017. PMID: 28949968 Free PMC article.
References
-
- Abdel Wahab N, Mason RM. Connective tissue growth factor and renal diseases: some answers, more questions. Curr Opin Nephrol Hypertens. 2004;13:53–58. - PubMed
-
- Boor P, Sebeková K, Ostendorf T, Floege J. Treatment targets in renal fibrosis. Nephrol Dial Transplant. 2007;22:3391–3407. - PubMed
-
- Bork P. The modular architecture of a new family of growth regulators related to connective tissue growth factor. FEBS Lett. 1993;327:125–130. - PubMed
-
- Chien W, Yin D, Gui D, Mori A, Frank JM, Said J, et al. Suppression of cell proliferation and signaling transduction by connective tissue growth factor in non-small cell lung cancer cells. Mol Cancer Res. 2006;4:591–598. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

