Metabolic consequences of mitochondrial coenzyme A deficiency in patients with PANK2 mutations
- PMID: 22221393
- PMCID: PMC3487396
- DOI: 10.1016/j.ymgme.2011.12.005
Metabolic consequences of mitochondrial coenzyme A deficiency in patients with PANK2 mutations
Abstract
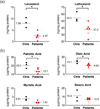
Pantothenate kinase-associated neurodegeneration (PKAN) is a rare, inborn error of metabolism characterized by iron accumulation in the basal ganglia and by the presence of dystonia, dysarthria, and retinal degeneration. Mutations in pantothenate kinase 2 (PANK2), the rate-limiting enzyme in mitochondrial coenzyme A biosynthesis, represent the most common genetic cause of this disorder. How mutations in this core metabolic enzyme give rise to such a broad clinical spectrum of pathology remains a mystery. To systematically explore its pathogenesis, we performed global metabolic profiling on plasma from a cohort of 14 genetically defined patients and 18 controls. Notably, lactate is elevated in PKAN patients, suggesting dysfunctional mitochondrial metabolism. As predicted, but never previously reported, pantothenate levels are higher in patients with premature stop mutations in PANK2. Global metabolic profiling and follow-up studies in patient-derived fibroblasts also reveal defects in bile acid conjugation and lipid metabolism, pathways that require coenzyme A. These findings raise a novel therapeutic hypothesis, namely, that dietary fats and bile acid supplements may hold potential as disease-modifying interventions. Our study illustrates the value of metabolic profiling as a tool for systematically exploring the biochemical basis of inherited metabolic diseases.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures






References
-
- Kurian MA, McNeill A, Lin JP, Maher ER. Childhood disorders of neurodegeneration with brain iron accumulation (NBIA) Dev. Med. Child. Neurol. 2011;53:394–404. - PubMed
-
- Spector R, Johanson CE. Vitamin transport and homeostasis in mammalian brain:focus on Vitamins B and E. J. Neurochem. 2007;103:425–438. - PubMed
-
- Daugherty M, Polanuyer B, Farrell M, Scholle M, Lykidis A, De Crécy-Lagard V, Osterman A. Complete reconstitution of the human coenzyme A biosynthetic pathway via comparative genomics. J. Biol. Chem. 2002;77:21431–21439. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

