Identity of the exchangeable sulfur-containing ligand at the Mo(V) center of R160Q human sulfite oxidase
- PMID: 22225516
- PMCID: PMC3273672
- DOI: 10.1021/ic201643t
Identity of the exchangeable sulfur-containing ligand at the Mo(V) center of R160Q human sulfite oxidase
Abstract
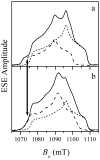
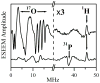
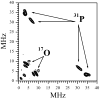
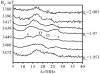
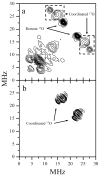
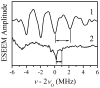
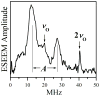
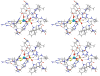
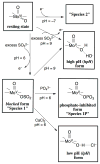
In our previous study of the fatal R160Q mutant of human sulfite oxidase (hSO) at low pH (Astashkin et al. J. Am. Chem. Soc.2008, 130, 8471-8480), a new Mo(V) species, denoted "species 1", was observed at low pH values. Species 1 was ascribed to a six-coordinate Mo(V) center with an exchangeable terminal oxo ligand and an equatorial sulfate group on the basis of pulsed EPR spectroscopy and (33)S and (17)O labeling. Here we report new results for species 1 of R160Q, based on substitution of the sulfur-containing ligand by a phosphate group, pulsed EPR spectroscopy in K(a)- and W-bands, and extensive density functional theory (DFT) calculations applied to large, more realistic molecular models of the enzyme active site. The combined results unambiguously show that species 1 has an equatorial sulfite as the only exchangeable ligand. The two types of (17)O signals that are observed arise from the coordinated and remote oxygen atoms of the sulfite ligand. A typical five-coordinate Mo(V) site is compatible with the observed and calculated EPR parameters.
Figures

Similar articles
-
Pulsed electron paramagnetic resonance spectroscopy of (33)S-labeled molybdenum cofactor in catalytically active bioengineered sulfite oxidase.Inorg Chem. 2014 Jan 21;53(2):961-71. doi: 10.1021/ic4023954. Epub 2014 Jan 3. Inorg Chem. 2014. PMID: 24387640 Free PMC article.
-
Structural studies of the molybdenum center of the pathogenic R160Q mutant of human sulfite oxidase by pulsed EPR spectroscopy and 17O and 33S labeling.J Am Chem Soc. 2008 Jul 2;130(26):8471-80. doi: 10.1021/ja801406f. Epub 2008 Jun 5. J Am Chem Soc. 2008. PMID: 18529001 Free PMC article.
-
Structure of the molybdenum site in YedY, a sulfite oxidase homologue from Escherichia coli.Inorg Chem. 2011 Feb 7;50(3):741-8. doi: 10.1021/ic101291j. Epub 2010 Dec 29. Inorg Chem. 2011. PMID: 21190337
-
Investigation of the coordination structures of the molybdenum(v) sites of sulfite oxidizing enzymes by pulsed EPR spectroscopy.Dalton Trans. 2006 Aug 7;(29):3501-14. doi: 10.1039/b602919a. Epub 2006 Jun 22. Dalton Trans. 2006. PMID: 16855750 Review.
-
Chemical systems modeling the d1 Mo(V) states of molybdenum enzymes.J Inorg Biochem. 2016 Sep;162:238-252. doi: 10.1016/j.jinorgbio.2016.06.010. Epub 2016 Jun 4. J Inorg Biochem. 2016. PMID: 27432259 Review.
Cited by
-
In vitro sulfuration of Rhodobacter capsulatus formate dehydrogenase.J Biol Chem. 2025 Jun;301(6):108511. doi: 10.1016/j.jbc.2025.108511. Epub 2025 Apr 15. J Biol Chem. 2025. PMID: 40246024 Free PMC article.
-
Mn(2+)-nucleotide coordination at the myosin active site as detected by pulsed electron paramagnetic resonance.J Phys Chem B. 2012 Nov 26;116(46):13655-62. doi: 10.1021/jp308423x. Epub 2012 Nov 13. J Phys Chem B. 2012. PMID: 23121488 Free PMC article.
-
Direct Spectroscopic Detection of ATP Turnover Reveals Mechanistic Divergence of ABC Exporters.Structure. 2017 Aug 1;25(8):1264-1274.e3. doi: 10.1016/j.str.2017.06.014. Epub 2017 Jul 14. Structure. 2017. PMID: 28712805 Free PMC article.
-
The mononuclear molybdenum enzymes.Chem Rev. 2014 Apr 9;114(7):3963-4038. doi: 10.1021/cr400443z. Epub 2014 Jan 28. Chem Rev. 2014. PMID: 24467397 Free PMC article. Review. No abstract available.
-
Pulsed electron paramagnetic resonance spectroscopy of (33)S-labeled molybdenum cofactor in catalytically active bioengineered sulfite oxidase.Inorg Chem. 2014 Jan 21;53(2):961-71. doi: 10.1021/ic4023954. Epub 2014 Jan 3. Inorg Chem. 2014. PMID: 24387640 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

