Synthesis of a γ-lactam library via formal cycloaddition of imines and substituted succinic anhydrides
- PMID: 22225535
- PMCID: PMC3325288
- DOI: 10.1021/co2001873
Synthesis of a γ-lactam library via formal cycloaddition of imines and substituted succinic anhydrides
Abstract
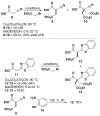
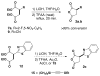
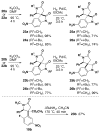
Formal cycloaddition reactions between imines and cyclic anhydrides serve as starting point for the synthesis of diverse libraries of small molecules. The synthesis of succinic anhydrides substituted with electron-withdrawing groups is facilitated by new mild conditions for alkylation of aryl-substituted acetyl esters with ethyl bromoacetate. These anhydrides are then used in formal cycloaddition reactions with imines to produce γ-lactams. 2-Fluoro-5-nitrophenylsuccinic anhydride reacts efficiently with imines to provide lactams that are further diversified by conversion of the nitro group to either an aniline and an azide for subsequent reactions with acylating agents and alkynes, respectively. The synthesis of cyanosuccinic anhydride is reported for the first time, and the use of this compound in reactions with imines and subsequent functionalization of the resultant lactams is demonstrated.
Figures
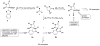
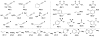
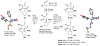

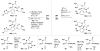
Similar articles
-
Diastereoselective synthesis of γ- and δ-lactams from imines and sulfone-substituted anhydrides.J Org Chem. 2014 Mar 21;79(6):2601-10. doi: 10.1021/jo500050n. Epub 2014 Mar 7. J Org Chem. 2014. PMID: 24552208 Free PMC article.
-
Stereoselective synthesis of γ-lactams from imines and cyanosuccinic anhydrides.Org Lett. 2013 Oct 4;15(19):5126-9. doi: 10.1021/ol402554n. Epub 2013 Sep 26. Org Lett. 2013. PMID: 24070117 Free PMC article.
-
Stereocontrol in asymmetric γ-lactam syntheses from imines and cyanosuccinic anhydrides.Org Lett. 2013 Oct 4;15(19):5130-3. doi: 10.1021/ol402561q. Epub 2013 Sep 26. Org Lett. 2013. PMID: 24070216 Free PMC article.
-
Cycloaddition reactions of imines with 3-thiosuccinic anhydrides: synthesis of the tricyclic core of martinellic acid.Org Lett. 2006 Aug 31;8(18):3999-4002. doi: 10.1021/ol061473z. Org Lett. 2006. PMID: 16928058
-
Trifluoromethylated lactams: promising small molecules in the search for effective drugs.Chem Commun (Camb). 2025 Jan 9;61(5):785-802. doi: 10.1039/d4cc05324a. Chem Commun (Camb). 2025. PMID: 39659174 Review.
Cited by
-
A construction of 4,4-spirocyclic γ-lactams by tandem radical cyclization with carbon monoxide.Beilstein J Org Chem. 2013 Jul 5;9:1340-5. doi: 10.3762/bjoc.9.151. eCollection 2013. Beilstein J Org Chem. 2013. PMID: 23946829 Free PMC article.
-
The Castagnoli-Cushman Reaction.Molecules. 2023 Mar 15;28(6):2654. doi: 10.3390/molecules28062654. Molecules. 2023. PMID: 36985626 Free PMC article. Review.
-
Angular-Substituted [1,4]Thiazino[3,4-a]Isoquinolines: Biological Evaluation and In Silico Studies on DPP-IV Inhibition.Int J Mol Sci. 2024 Nov 1;25(21):11753. doi: 10.3390/ijms252111753. Int J Mol Sci. 2024. PMID: 39519303 Free PMC article.
-
Diastereoselective synthesis of γ- and δ-lactams from imines and sulfone-substituted anhydrides.J Org Chem. 2014 Mar 21;79(6):2601-10. doi: 10.1021/jo500050n. Epub 2014 Mar 7. J Org Chem. 2014. PMID: 24552208 Free PMC article.
-
Synthesis of a library of "lead-like" γ-lactams by a one pot, four-component reaction.ACS Comb Sci. 2013 Jul 8;15(7):356-62. doi: 10.1021/co400049f. Epub 2013 Jun 12. ACS Comb Sci. 2013. PMID: 23682712 Free PMC article.
References
-
- Mori T, Takahashi K, Kashiwabara M, Uemura D, Katayama C, Iwadare S, Shizuri Y, Mitomo R, Nakano F, Matsuzaki A. Structure of oxazolomycin, a novel β-lactone antibiotic. Tetrahedron Lett. 1985;26:1073–1076.
-
- Feling RH, Buchanan GO, Mincer TJ, Kauffman CA, Jensen PR, Fenical W. Salinosporamide a: A highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus salinospora. Angew Chem, Int Ed. 2003;42:355–357. - PubMed
-
- Guntern A, Ioset JR, Queiroz EF, Sandor P, Foggin CM, Hostettmann K. Heliotropamide, a novel oxopyrrolidine-3-carboxamide from heliotropium ovalifolium. J Nat Prod. 2003;66:1550–1553. - PubMed
-
- Manam RR, Teisan S, White DJ, Nicholson B, Grodberg J, Neuteboom STC, Lam KS, Mosca DA, Lloyd GK, Potts BCM. Lajollamycin, a nitro-tetraene spiro-β-lactone-γ-lactam antibiotic from the marine actinomycete streptomyces nodosus. J Nat Prod. 2005;68:240–243. - PubMed
-
- Okazaki Y, Ishizuka A, Ishihara A, Nishioka T, Iwamura H. New dimeric compounds of avenanthramide phytoalexin in oats. J Org Chem. 2007;72:3830–3839. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

