Collective and single cell behavior in epithelial contact inhibition
- PMID: 22228306
- PMCID: PMC3271933
- DOI: 10.1073/pnas.1007809109
Collective and single cell behavior in epithelial contact inhibition
Abstract
Control of cell proliferation is a fundamental aspect of tissue physiology central to morphogenesis, wound healing, and cancer. Although many of the molecular genetic factors are now known, the system level regulation of growth is still poorly understood. A simple form of inhibition of cell proliferation is encountered in vitro in normally differentiating epithelial cell cultures and is known as "contact inhibition." The study presented here provides a quantitative characterization of contact inhibition dynamics on tissue-wide and single cell levels. Using long-term tracking of cultured Madin-Darby canine kidney cells we demonstrate that inhibition of cell division in a confluent monolayer follows inhibition of cell motility and sets in when mechanical constraint on local expansion causes divisions to reduce cell area. We quantify cell motility and cell cycle statistics in the low density confluent regime and their change across the transition to epithelial morphology which occurs with increasing cell density. We then study the dynamics of cell area distribution arising through reductive division, determine the average mitotic rate as a function of cell size, and demonstrate that complete arrest of mitosis occurs when cell area falls below a critical value. We also present a simple computational model of growth mechanics which captures all aspects of the observed behavior. Our measurements and analysis show that contact inhibition is a consequence of mechanical interaction and constraint rather than interfacial contact alone, and define quantitative phenotypes that can guide future studies of molecular mechanisms underlying contact inhibition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
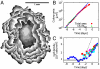

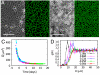
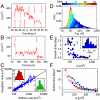
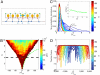
Comment in
-
Re: Collective and single cell behavior in epithelial contact inhibition.J Urol. 2012 Oct;188(4):1396-7. doi: 10.1016/j.juro.2012.06.073. Epub 2012 Aug 18. J Urol. 2012. PMID: 22971414 No abstract available.
References
-
- Leevers SJ, McNeill H. Controlling the size of organs and organisms. Curr Opin Cell Biol. 2005;17:604–609. - PubMed
-
- Lecuit T, Le Goff L. Orchestrating size and shape during morphogenesis. Nature. 2007;450:189–192. - PubMed
-
- Alberts B, et al. Molecular Biology of the Cell. New York: Garland; 2008.
-
- Chaffer CL, Thompson EW, Williams ED. Mesenchymal to epithelial transition in development and disease. Cells Tissues Organs. 2007;185:7–19. - PubMed
-
- Guarino M, Micheli P, Pallotti F, Giordano F. Pathological relevance of epithelial and mesenchymal phenotype plasticity. Pathol Res Pract. 1999;195:379–89. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

