Three-dimensional structures of pathogenic and saprophytic Leptospira species revealed by cryo-electron tomography
- PMID: 22228733
- PMCID: PMC3294836
- DOI: 10.1128/JB.06474-11
Three-dimensional structures of pathogenic and saprophytic Leptospira species revealed by cryo-electron tomography
Abstract
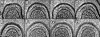
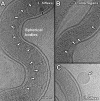
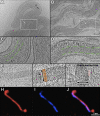
Leptospira interrogans is the primary causative agent of the most widespread zoonotic disease, leptospirosis. An in-depth structural characterization of L. interrogans is needed to understand its biology and pathogenesis. In this study, cryo-electron tomography (cryo-ET) was used to compare pathogenic and saprophytic species and examine the unique morphological features of this group of bacteria. Specifically, our study revealed a structural difference between the cell envelopes of L. interrogans and Leptospira biflexa involving variations in the lipopolysaccharide (LPS) layer. Through cryo-ET and subvolume averaging, we determined the first three-dimensional (3-D) structure of the flagellar motor of leptospira, with novel features in the flagellar C ring, export apparatus, and stator. Together with direct visualization of chemoreceptor arrays, DNA packing, periplasmic filaments, spherical cytoplasmic bodies, and a unique "cap" at the cell end, this report provides structural insights into these fascinating Leptospira species.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

