Cardiac aging: from molecular mechanisms to significance in human health and disease
- PMID: 22229339
- PMCID: PMC3329953
- DOI: 10.1089/ars.2011.4179
Cardiac aging: from molecular mechanisms to significance in human health and disease
Abstract
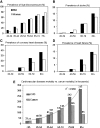
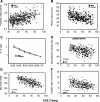
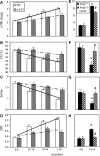
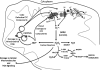
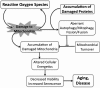
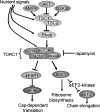
Cardiovascular diseases (CVDs) are the major causes of death in the western world. The incidence of cardiovascular disease as well as the rate of cardiovascular mortality and morbidity increase exponentially in the elderly population, suggesting that age per se is a major risk factor of CVDs. The physiologic changes of human cardiac aging mainly include left ventricular hypertrophy, diastolic dysfunction, valvular degeneration, increased cardiac fibrosis, increased prevalence of atrial fibrillation, and decreased maximal exercise capacity. Many of these changes are closely recapitulated in animal models commonly used in an aging study, including rodents, flies, and monkeys. The application of genetically modified aged mice has provided direct evidence of several critical molecular mechanisms involved in cardiac aging, such as mitochondrial oxidative stress, insulin/insulin-like growth factor/PI3K pathway, adrenergic and renin angiotensin II signaling, and nutrient signaling pathways. This article also reviews the central role of mitochondrial oxidative stress in CVDs and the plausible mechanisms underlying the progression toward heart failure in the susceptible aging hearts. Finally, the understanding of the molecular mechanisms of cardiac aging may support the potential clinical application of several "anti-aging" strategies that treat CVDs and improve healthy cardiac aging.
Figures




References
-
- Adlam VJ. Harrison JC. Porteous CM. James AM. Smith RA. Murphy MP. Sammut IA. Targeting an antioxidant to mitochondria decreases cardiac ischemia-reperfusion injury. Faseb J. 2005;19:1088–1095. - PubMed
-
- Ahmed HM. Blaha MJ. Nasir K. Rivera JJ. Blumenthal RS. Effects of physical activity on cardiovascular disease. Am J Cardiol. 2012;109:288–295. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

