Mechanisms of action of escapin, a bactericidal agent in the ink secretion of the sea hare Aplysia californica: rapid and long-lasting DNA condensation and involvement of the OxyR-regulated oxidative stress pathway
- PMID: 22232273
- PMCID: PMC3318362
- DOI: 10.1128/AAC.05874-11
Mechanisms of action of escapin, a bactericidal agent in the ink secretion of the sea hare Aplysia californica: rapid and long-lasting DNA condensation and involvement of the OxyR-regulated oxidative stress pathway
Abstract
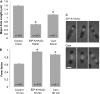
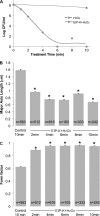
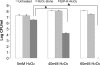
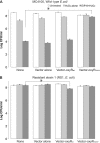
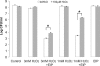
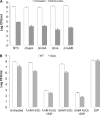
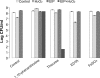
The marine snail Aplysia californica produces escapin, an L-amino acid oxidase, in its defensive ink. Escapin uses L-lysine to produce diverse products called escapin intermediate products of L-lysine (EIP-K), including α-amino-ε-caproic acid, Δ¹-piperidine-2-carboxylic acid, and Δ²-piperidine-2-carboxylic acid. EIP-K and H₂O₂ together, but neither alone, is a powerful bactericide. Here, we report bactericidal mechanisms of escapin products on Escherichia coli. We show that EIP-K and H₂O₂ together cause rapid and long-lasting DNA condensation: 2-min treatment causes significant DNA condensation and killing, and 10-min treatment causes maximal effect, lasting at least 70 h. We isolated two mutants resistant to EIP-K plus H₂O₂, both having a single missense mutation in the oxidation regulatory gene, oxyR. A complementation assay showed that the mutated gene, oxyR(A233V), renders resistance to EIP-K plus H₂O₂, and a gene dosage effect leads to reduction of resistance for strains carrying wild-type oxyR. Temperature stress with EIP-K does not produce the bactericidal effect, suggesting the effect is due to a specific response to oxidative stress. The null mutant for any single DNA-binding protein--Dps, H-NS, Hup, Him, or MukB--was not resistant to EIP-K plus H₂O₂, suggesting that no single DNA-binding protein is necessary to mediate this bactericidal effect, but allowing for the possibility that EIP-K plus H₂O₂ could function through a combination of DNA-binding proteins. The bactericidal effect of EIP-K plus H₂O₂ was eliminated by the ferrous ion chelator 1,10-phenanthroline, and it was reduced by the hydroxyl radical scavenger thiourea, suggesting hydroxyl radicals mediate the effects of EIP-K plus H₂O₂.
Figures


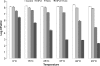
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

