Neurotrophic factor changes in the rat thick skin following chronic constriction injury of the sciatic nerve
- PMID: 22233577
- PMCID: PMC3269365
- DOI: 10.1186/1744-8069-8-1
Neurotrophic factor changes in the rat thick skin following chronic constriction injury of the sciatic nerve
Abstract
Background: Cutaneous peripheral neuropathies have been associated with changes of the sensory fiber innervation in the dermis and epidermis. These changes are mediated in part by the increase in local expression of trophic factors. Increase in target tissue nerve growth factor has been implicated in the promotion of peptidergic afferent and sympathetic efferent sprouting following nerve injury. The primary source of nerve growth factor is cells found in the target tissue, namely the skin. Recent evidence regarding the release and extracellular maturation of nerve growth factor indicate that it is produced in its precursor form and matured in the extracellular space. It is our hypothesis that the precursor form of nerve growth factor should be detectable in those cell types producing it. To date, limitations in available immunohistochemical tools have restricted efforts in obtaining an accurate distribution of nerve growth factor in the skin of naïve animals and those with neuropathic pain lesions. It is the objective of this study to delineate the distribution of the precursor form of nerve growth factor to those cell types expressing it, as well as to describe its distribution with respect to those nerve fibers responsive to it.
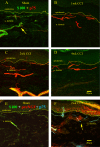
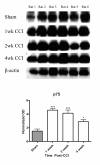
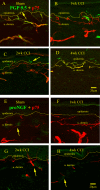
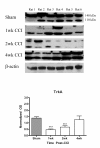
Results: We observed a decrease in peptidergic fiber innervation at 1 week after the application of a chronic constriction injury (CCI) to the sciatic nerve, followed by a recovery, correlating with TrkA protein levels. ProNGF expression in CCI animals was significantly higher than in sham-operated controls from 1-4 weeks post-CCI. ProNGF immunoreactivity was increased in mast cells at 1 week post-CCI and, at later time points, in keratinocytes. P75 expression within the dermis and epidermis was significantly higher in CCI-operated animals than in controls and these changes were localized to neuronal and non-neuronal cell populations using specific markers for each.
Conclusions: We describe proNGF expression by non-neuronal cells over time after nerve injury as well as the association of NGF-responsive fibers to proNGF-expressing target tissues. ProNGF expression increases following nerve injury in those cell types previously suggested to express it.
Figures


Similar articles
-
Delayed reinnervation by nonpeptidergic nociceptive afferents of the glabrous skin of the rat hindpaw in a neuropathic pain model.J Comp Neurol. 2011 Jan 1;519(1):49-63. doi: 10.1002/cne.22500. J Comp Neurol. 2011. PMID: 21120927
-
Redistribution of voltage-gated sodium channels after nerve decompression contributes to relieve neuropathic pain in chronic constriction injury.Brain Res. 2014 Nov 17;1589:15-25. doi: 10.1016/j.brainres.2014.07.012. Epub 2014 Jul 16. Brain Res. 2014. PMID: 25038561
-
Autonomic fibre sprouting and changes in nociceptive sensory innervation in the rat lower lip skin following chronic constriction injury.Eur J Neurosci. 2005 May;21(9):2475-87. doi: 10.1111/j.1460-9568.2005.04089.x. Eur J Neurosci. 2005. PMID: 15932605
-
NGF and ProNGF: Regulation of neuronal and neoplastic responses through receptor signaling.Adv Biol Regul. 2015 May;58:16-27. doi: 10.1016/j.jbior.2014.11.003. Epub 2014 Nov 20. Adv Biol Regul. 2015. PMID: 25491371 Free PMC article. Review.
-
Distribution of the tetrodotoxin-resistant sodium channel PN3 in rat sensory neurons in normal and neuropathic conditions.J Neurosci. 1998 Mar 15;18(6):2174-87. doi: 10.1523/JNEUROSCI.18-06-02174.1998. J Neurosci. 1998. PMID: 9482802 Free PMC article. Review.
Cited by
-
New topical treatment of vulvodynia based on the pathogenetic role of cross talk between nociceptors, immunocompetent cells, and epithelial cells.J Pain Res. 2016 Oct 3;9:757-762. doi: 10.2147/JPR.S115407. eCollection 2016. J Pain Res. 2016. PMID: 27757050 Free PMC article.
-
Physiologic facet capsule stretch can induce pain & upregulate matrix metalloproteinase-3 in the dorsal root ganglia when preceded by a physiological mechanical or nonpainful chemical exposure.Clin Biomech (Bristol). 2019 Apr;64:122-130. doi: 10.1016/j.clinbiomech.2018.01.009. Epub 2018 Jan 31. Clin Biomech (Bristol). 2019. PMID: 29523370 Free PMC article.
-
Schwann cells use TAM receptor-mediated phagocytosis in addition to autophagy to clear myelin in a mouse model of nerve injury.Proc Natl Acad Sci U S A. 2017 Sep 19;114(38):E8072-E8080. doi: 10.1073/pnas.1710566114. Epub 2017 Sep 5. Proc Natl Acad Sci U S A. 2017. PMID: 28874532 Free PMC article.
-
Removal of half the sympathetic innervation does not reduce vasoconstrictor responses in rat tail artery.J Physiol. 2013 Jun 1;591(11):2867-84. doi: 10.1113/jphysiol.2012.250365. Epub 2013 Apr 3. J Physiol. 2013. PMID: 23551946 Free PMC article.
-
Pro-neurotrophins, sortilin, and nociception.Eur J Neurosci. 2014 Feb;39(3):363-74. doi: 10.1111/ejn.12466. Eur J Neurosci. 2014. PMID: 24494677 Free PMC article. Review.
References
-
- Levi-Montalcini R, Angeletti PU. Essential role of the nerve growth factor on the survival and maintenance of dissociated sensory and sympathetic embryonic nerve cells in vitro. Dev Biol. 1963;7:653–659. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

