Dynamin, a membrane-remodelling GTPase
- PMID: 22233676
- PMCID: PMC3519936
- DOI: 10.1038/nrm3266
Dynamin, a membrane-remodelling GTPase
Abstract
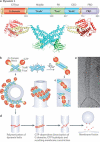
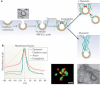
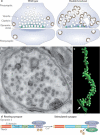
Dynamin, the founding member of a family of dynamin-like proteins (DLPs) implicated in membrane remodelling, has a critical role in endocytic membrane fission events. The use of complementary approaches, including live-cell imaging, cell-free studies, X-ray crystallography and genetic studies in mice, has greatly advanced our understanding of the mechanisms by which dynamin acts, its essential roles in cell physiology and the specific function of different dynamin isoforms. In addition, several connections between dynamin and human disease have also emerged, highlighting specific contributions of this GTPase to the physiology of different tissues.
Figures

References
-
- McMahon HT, Boucrot E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nature reviews. Molecular cell biology. 2011;12:517–33. - PubMed
-
- Mercer J, Schelhaas M, Helenius A. Virus entry by endocytosis. Annual review of biochemistry. 2010;79:803–33. - PubMed
-
- Scita G, Di Fiore PP. The endocytic matrix. Nature. 2010;463:464–73. - PubMed
-
- Howes MT, Mayor S, Parton RG. Molecules, mechanisms, and cellular roles of clathrin-independent endocytosis. Current opinion in cell biology. 2010;22:519–27. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

