Essential role for Stat5a/b in myeloproliferative neoplasms induced by BCR-ABL1 and JAK2(V617F) in mice
- PMID: 22234689
- PMCID: PMC3325042
- DOI: 10.1182/blood-2011-12-397554
Essential role for Stat5a/b in myeloproliferative neoplasms induced by BCR-ABL1 and JAK2(V617F) in mice
Abstract
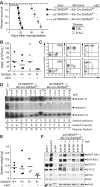
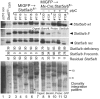
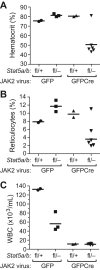
STAT5 proteins are constitutively activated in malignant cells from many patients with leukemia, including the myeloproliferative neoplasms (MPNs) chronic myeloid leukemia (CML) and polycythemia vera (PV), but whether STAT5 is essential for the pathogenesis of these diseases is not known. In the present study, we used mice with a conditional null mutation in the Stat5a/b gene locus to determine the requirement for STAT5 in MPNs induced by BCR-ABL1 and JAK2(V617F) in retroviral transplantation models of CML and PV. Loss of one Stat5a/b allele resulted in a decrease in BCR-ABL1-induced CML-like MPN and the appearance of B-cell acute lymphoblastic leukemia, whereas complete deletion of Stat5a/b prevented the development of leukemia in primary recipients. However, BCR-ABL1 was expressed and active in Stat5-null leukemic stem cells, and Stat5 deletion did not prevent progression to lymphoid blast crisis or abolish established B-cell acute lymphoblastic leukemia. JAK2(V617F) failed to induce polycythemia in recipients after deletion of Stat5a/b, although the loss of STAT5 did not prevent the development of myelofibrosis. These results demonstrate that STAT5a/b is essential for the induction of CML-like leukemia by BCR-ABL1 and of polycythemia by JAK2(V617F), and validate STAT5a/b and the genes they regulate as targets for therapy in these MPNs.
Figures




Comment in
-
Advancing the STATus of MPN pathogenesis.Blood. 2012 Apr 12;119(15):3374-6. doi: 10.1182/blood-2012-02-406611. Blood. 2012. PMID: 22500049
References
-
- Teglund S, McKay C, Schuetz E, et al. Stat5a and Stat5b proteins have essential and nonessential, or redundant, roles in cytokine responses. Cell. 1998;93(5):841–850. - PubMed
-
- Liu X, Robinson GW, Wagner KU, Garrett L, Wynshaw-Boris A, Hennignausen L. Stat5a is mandatory for adult mammary gland development and lactogenesis. Genes Dev. 1997;11(2):179–186. - PubMed
-
- Moriggl R, Sexl V, Kenner L, et al. Stat5 tetramer formation is associated with leukemogenesis. Cancer Cell. 2005;7(1):87–99. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

