Localized iron supply triggers lateral root elongation in Arabidopsis by altering the AUX1-mediated auxin distribution
- PMID: 22234997
- PMCID: PMC3289578
- DOI: 10.1105/tpc.111.092973
Localized iron supply triggers lateral root elongation in Arabidopsis by altering the AUX1-mediated auxin distribution
Abstract
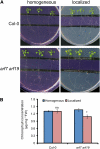
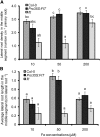
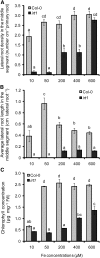
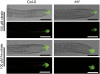
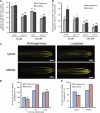
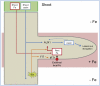
Root system architecture depends on nutrient availability, which shapes primary and lateral root development in a nutrient-specific manner. To better understand how nutrient signals are integrated into root developmental programs, we investigated the morphological response of Arabidopsis thaliana roots to iron (Fe). Relative to a homogeneous supply, localized Fe supply in horizontally separated agar plates doubled lateral root length without having a differential effect on lateral root number. In the Fe uptake-defective mutant iron-regulated transporter1 (irt1), lateral root development was severely repressed, but a requirement for IRT1 could be circumvented by Fe application to shoots, indicating that symplastic Fe triggered the local elongation of lateral roots. The Fe-stimulated emergence of lateral root primordia and root cell elongation depended on the rootward auxin stream and was accompanied by a higher activity of the auxin reporter DR5-β-glucuronidase in lateral root apices. A crucial role of the auxin transporter AUXIN RESISTANT1 (AUX1) in Fe-triggered lateral root elongation was indicated by Fe-responsive AUX1 promoter activities in lateral root apices and by the failure of the aux1-T mutant to elongate lateral roots into Fe-enriched agar patches. We conclude that a local symplastic Fe gradient in lateral roots upregulates AUX1 to accumulate auxin in lateral root apices as a prerequisite for lateral root elongation.
Figures
Similar articles
-
Auxin Influx Carrier AUX1 Confers Acid Resistance for Arabidopsis Root Elongation Through the Regulation of Plasma Membrane H+-ATPase.Plant Cell Physiol. 2016 Oct;57(10):2194-2201. doi: 10.1093/pcp/pcw136. Epub 2016 Aug 8. Plant Cell Physiol. 2016. PMID: 27503216 Free PMC article.
-
Shoot-supplied ammonium targets the root auxin influx carrier AUX1 and inhibits lateral root emergence in Arabidopsis.Plant Cell Environ. 2011 Jun;34(6):933-946. doi: 10.1111/j.1365-3040.2011.02295.x. Epub 2011 Mar 24. Plant Cell Environ. 2011. PMID: 21342208
-
Auxin and ethylene are involved in the responses of root system architecture to low boron supply in Arabidopsis seedlings.Physiol Plant. 2011 Jun;142(2):170-8. doi: 10.1111/j.1399-3054.2011.01459.x. Epub 2011 Mar 16. Physiol Plant. 2011. PMID: 21338369
-
Signalling Overlaps between Nitrate and Auxin in Regulation of The Root System Architecture: Insights from the Arabidopsis thaliana.Int J Mol Sci. 2020 Apr 20;21(8):2880. doi: 10.3390/ijms21082880. Int J Mol Sci. 2020. PMID: 32326090 Free PMC article. Review.
-
Lateral root initiation is a probabilistic event whose frequency is set by fluctuating levels of auxin response.J Exp Bot. 2013 Jun;64(9):2609-17. doi: 10.1093/jxb/ert155. J Exp Bot. 2013. PMID: 23709673 Review.
Cited by
-
Molecular and physiological control of adventitious rooting in cuttings: phytohormone action meets resource allocation.Ann Bot. 2019 Jun 24;123(6):929-949. doi: 10.1093/aob/mcy234. Ann Bot. 2019. PMID: 30759178 Free PMC article.
-
A specific role of iron in promoting meristematic cell division during adventitious root formation.J Exp Bot. 2017 Jul 10;68(15):4233-4247. doi: 10.1093/jxb/erx248. J Exp Bot. 2017. PMID: 28922771 Free PMC article.
-
Decoding Long-Distance Communication Under Mineral Stress: Advances in Vascular Signalling and Molecular Tools for Plant Resilience.Plant Cell Environ. 2025 Jul;48(7):4781-4802. doi: 10.1111/pce.15475. Epub 2025 Mar 17. Plant Cell Environ. 2025. PMID: 40091594 Free PMC article. Review.
-
Correlation analysis of proteins responsive to Zn, Mn, or Fe deficiency in Arabidopsis roots based on iTRAQ analysis.Plant Cell Rep. 2015 Jan;34(1):157-66. doi: 10.1007/s00299-014-1696-2. Epub 2014 Nov 1. Plant Cell Rep. 2015. PMID: 25366567
-
Plant microbiota controls an alternative root branching regulatory mechanism in plants.Proc Natl Acad Sci U S A. 2023 Apr 11;120(15):e2301054120. doi: 10.1073/pnas.2301054120. Epub 2023 Apr 3. Proc Natl Acad Sci U S A. 2023. PMID: 37011213 Free PMC article.
References
-
- Bao J., Chen F.J., Gu R.L., Wang G.Y., Zhang F.S., Mi G.H. (2007). Lateral root development of two Arabidopsis auxin transport mutants, aux1-7 and eir1-1, in response to nitrate supplies. Plant Sci. 173: 417–425
-
- Bauer P., Ling H.Q., Guerinot M.L. (2007). FIT, the FER-LIKE IRON DEFICIENCY INDUCED TRANSCRIPTION FACTOR in Arabidopsis. Plant Physiol. Biochem. 45: 260–261 - PubMed
-
- Casimiro I., Beeckman T., Graham N., Bhalerao R., Zhang H., Casero P., Sandberg G., Bennett M.J. (2003). Dissecting Arabidopsis lateral root development. Trends Plant Sci. 8: 165–171 - PubMed
-
- Chaney R.L. (1988). Plants can utilize iron from Fe-N,N'-Di-(2-hydroxybenzoyl)-ethylenediamine-N,N'-diacetic acid, a ferric chelate with 106 greater formation constant than Fe-Eddha. J. Plant Nutr. 11: 1033–1050
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

