The cycling of acetyl-coenzyme A through acetylcarnitine buffers cardiac substrate supply: a hyperpolarized 13C magnetic resonance study
- PMID: 22238215
- PMCID: PMC3378498
- DOI: 10.1161/CIRCIMAGING.111.969451
The cycling of acetyl-coenzyme A through acetylcarnitine buffers cardiac substrate supply: a hyperpolarized 13C magnetic resonance study
Abstract
Background: Carnitine acetyltransferase catalyzes the reversible conversion of acetyl-coenzyme A (CoA) into acetylcarnitine. The aim of this study was to use the metabolic tracer hyperpolarized [2-(13)C]pyruvate with magnetic resonance spectroscopy to determine whether carnitine acetyltransferase facilitates carbohydrate oxidation in the heart.
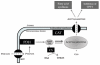
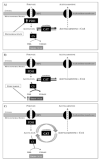
Methods and results: Ex vivo, following hyperpolarized [2-(13)C]pyruvate infusion, the [1-(13)C]acetylcarnitine resonance was saturated with a radiofrequency pulse, and the effect of this saturation on [1-(13)C]citrate and [5-(13)C]glutamate was observed. In vivo, [2-(13)C]pyruvate was infused into 3 groups of fed male Wistar rats: (1) controls, (2) rats in which dichloroacetate enhanced pyruvate dehydrogenase flux, and (3) rats in which dobutamine elevated cardiac workload. In the perfused heart, [1-(13)C]acetylcarnitine saturation reduced the [1-(13)C]citrate and [5-(13)C]glutamate resonances by 63% and 51%, respectively, indicating a rapid exchange between pyruvate-derived acetyl-CoA and the acetylcarnitine pool. In vivo, dichloroacetate increased the rate of [1-(13)C]acetylcarnitine production by 35% and increased the overall acetylcarnitine pool size by 33%. Dobutamine decreased the rate of [1-(13)C]acetylcarnitine production by 37% and decreased the acetylcarnitine pool size by 40%.
Conclusions: Hyperpolarized (13)C magnetic resonance spectroscopy has revealed that acetylcarnitine provides a route of disposal for excess acetyl-CoA and a means to replenish acetyl-CoA when cardiac workload is increased. Cycling of acetyl-CoA through acetylcarnitine appears key to matching instantaneous acetyl-CoA supply with metabolic demand, thereby helping to balance myocardial substrate supply and contractile function.
Figures




Comment in
-
In vivo clinical measures of intermediary metabolism are inadequate: can a new magnetic resonance spectroscopy technology do better?Circ Cardiovasc Imaging. 2012 Mar;5(2):171-4. doi: 10.1161/CIRCIMAGING.111.972240. Circ Cardiovasc Imaging. 2012. PMID: 22438422 No abstract available.
Similar articles
-
Multiple mass isotopomer tracing of acetyl-CoA metabolism in Langendorff-perfused rat hearts: channeling of acetyl-CoA from pyruvate dehydrogenase to carnitine acetyltransferase.J Biol Chem. 2015 Mar 27;290(13):8121-32. doi: 10.1074/jbc.M114.631549. Epub 2015 Feb 2. J Biol Chem. 2015. PMID: 25645937 Free PMC article.
-
In vivo clinical measures of intermediary metabolism are inadequate: can a new magnetic resonance spectroscopy technology do better?Circ Cardiovasc Imaging. 2012 Mar;5(2):171-4. doi: 10.1161/CIRCIMAGING.111.972240. Circ Cardiovasc Imaging. 2012. PMID: 22438422 No abstract available.
-
In vivo investigation of cardiac metabolism in the rat using MRS of hyperpolarized [1-13C] and [2-13C]pyruvate.NMR Biomed. 2013 Dec;26(12):1680-7. doi: 10.1002/nbm.3003. Epub 2013 Jul 31. NMR Biomed. 2013. PMID: 23904148 Free PMC article.
-
Regulation by carnitine of myocardial fatty acid and carbohydrate metabolism under normal and pathological conditions.Basic Res Cardiol. 2000 Apr;95(2):75-83. doi: 10.1007/s003950050167. Basic Res Cardiol. 2000. PMID: 10826498 Review.
-
Clinical implications of cardiac hyperpolarized magnetic resonance imaging.J Cardiovasc Magn Reson. 2013 Oct 8;15(1):93. doi: 10.1186/1532-429X-15-93. J Cardiovasc Magn Reson. 2013. PMID: 24103786 Free PMC article. Review.
Cited by
-
Carnitine Acetyltransferase Mitigates Metabolic Inertia and Muscle Fatigue during Exercise.Cell Metab. 2015 Jul 7;22(1):65-76. doi: 10.1016/j.cmet.2015.06.003. Cell Metab. 2015. PMID: 26154055 Free PMC article.
-
Spontaneous hydrolysis and spurious metabolic properties of α-ketoglutarate esters.Nat Commun. 2021 Aug 12;12(1):4905. doi: 10.1038/s41467-021-25228-9. Nat Commun. 2021. PMID: 34385458 Free PMC article.
-
Imaging oxygen metabolism with hyperpolarized magnetic resonance: a novel approach for the examination of cardiac and renal function.Biosci Rep. 2017 Jan 27;37(1):BSR20160186. doi: 10.1042/BSR20160186. Print 2017 Feb 28. Biosci Rep. 2017. PMID: 27899435 Free PMC article. Review.
-
Acetate Revisited: A Key Biomolecule at the Nexus of Metabolism, Epigenetics and Oncogenesis-Part 1: Acetyl-CoA, Acetogenesis and Acyl-CoA Short-Chain Synthetases.Front Physiol. 2020 Nov 12;11:580167. doi: 10.3389/fphys.2020.580167. eCollection 2020. Front Physiol. 2020. PMID: 33281616 Free PMC article. Review.
-
Measuring changes in substrate utilization in the myocardium in response to fasting using hyperpolarized [1-(13)C]butyrate and [1-(13)C]pyruvate.Sci Rep. 2016 May 6;6:25573. doi: 10.1038/srep25573. Sci Rep. 2016. PMID: 27150735 Free PMC article.
References
-
- Hansford RG, Cohen L. Relative importance of pyruvate dehydrogenase interconversion and feed-back inhibition in the effect of fatty acids on pyruvate oxidation by rat heart mitochondria. Arch Biochem Biophys. 1978;191:65–81. - PubMed
-
- Collins-Nakai RL, Noseworthy D, Lopaschuk GD. Epinephrine increases atp production in hearts by preferentially increasing glucose metabolism. The American journal of physiology. 1994;267:H1862–1871. - PubMed
-
- Owen OE, Mozzoli MA, Boden G, Patel MS, Reichard GA, Jr., Trapp V, Shuman CR, Felig P. Substrate, hormone, and temperature responses in males and females to a common breakfast. Metabolism. 1980;29:511–523. - PubMed
-
- Cross HR, Opie LH, Radda GK, Clarke K. Is a high glycogen content beneficial or detrimental to the ischemic rat heart? A controversy resolved. Circulation research. 1996;78:482–491. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

