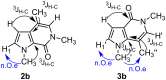Access to pyrrolo-pyridines by gold-catalyzed hydroarylation of pyrroles tethered to terminal alkynes
- PMID: 22238519
- PMCID: PMC3252845
- DOI: 10.3762/bjoc.7.170
Access to pyrrolo-pyridines by gold-catalyzed hydroarylation of pyrroles tethered to terminal alkynes
Abstract
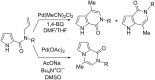
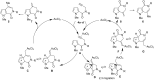
In a simple procedure, the intramolecular hydroarylation of N-propargyl-pyrrole-2-carboxamides was accomplished with the aid of gold(III) catalysis. The reaction led to differently substituted pyrrolo[2,3-c]pyridine and pyrrolo[3,2-c]pyridine derivatives arising either from direct cyclization or from a formal rearrangement of the carboxamide group. Terminal alkynes are essential to achieve bicyclic pyrrolo-fused pyridinones by a 6-exo-dig process, while the presence of a phenyl group at the C-C triple bond promotes the 7-endo-dig cyclization giving pyrrolo-azepines.
Keywords: C–C coupling; gold catalysis; homogeneous catalysis; nitrogen heterocycles; rearrangement.
Figures
Similar articles
-
Gold-catalyzed oxime-oxime rearrangement.Org Lett. 2015 Jun 5;17(11):2660-3. doi: 10.1021/acs.orglett.5b01041. Epub 2015 May 20. Org Lett. 2015. PMID: 25992473
-
Intramolecular Gold-Catalyzed and NaH-Supported Cyclization Reactions of N-Propargyl Indole Derivatives with Pyrazole and Pyrrole Rings: Synthesis of Pyrazolodiazepinoindole, Pyrazolopyrazinoindole, and Pyrrolopyrazinoindole.J Org Chem. 2015 Dec 18;80(24):12552-61. doi: 10.1021/acs.joc.5b02419. Epub 2015 Dec 8. J Org Chem. 2015. PMID: 26629889
-
Palladium-catalyzed Sonogashira-coupling conjoined C-H activation: a regioselective tandem strategy to access indolo- and pyrrolo[1,2-a]quinolines.J Org Chem. 2012 Nov 16;77(22):10382-92. doi: 10.1021/jo302112a. Epub 2012 Oct 30. J Org Chem. 2012. PMID: 23083404
-
An Overview of the Biological Activity of Pyrrolo[3,4-c]pyridine Derivatives.Pharmaceuticals (Basel). 2021 Apr 11;14(4):354. doi: 10.3390/ph14040354. Pharmaceuticals (Basel). 2021. PMID: 33920479 Free PMC article. Review.
-
Ring strain strategy for the control of regioselectivity. Gold-catalyzed anti-Markovnikov cycloisomerization initiated tandem reactions of alkynes.Sci Bull (Beijing). 2017 Mar 1;62(5):352-357. doi: 10.1016/j.scib.2017.01.016. Epub 2017 Jan 18. Sci Bull (Beijing). 2017. PMID: 36659420 Review.
Cited by
-
Merging gold and organocatalysis: a facile asymmetric synthesis of annulated pyrroles.Chemistry. 2014 Apr 1;20(14):3917-21. doi: 10.1002/chem.201400407. Epub 2014 Mar 3. Chemistry. 2014. PMID: 24590817 Free PMC article.
References
-
- Li J J, Gribble G W. Palladium in Heterocyclic Chemistry. A Guide for the Synthetic Chemist. New York: Pergamon; 2000.
-
- Beccalli E M, Broggini G, Fasana A, Rigamonti M. J Organomet Chem. 2011;696:277–295. doi: 10.1016/j.jorganchem.2010.09.078. - DOI
LinkOut - more resources
Full Text Sources