Unveiling the mechanisms for decreased glutathione in individuals with HIV infection
- PMID: 22242038
- PMCID: PMC3254057
- DOI: 10.1155/2012/734125
Unveiling the mechanisms for decreased glutathione in individuals with HIV infection
Abstract
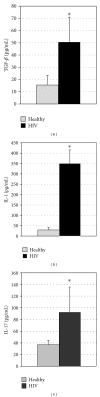
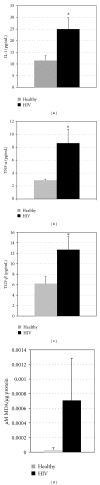
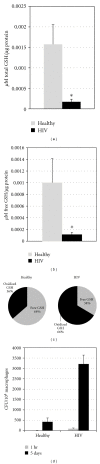
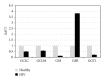
We examined the causes for decreased glutathione (GSH) in individuals with HIV infection. We observed lower levels of intracellular GSH in macrophages from individuals with HIV compared to healthy subjects. Further, the GSH composition found in macrophages from HIV(+) subjects heavily favors oxidized glutathione (GSSG) which lacks antioxidant activity, over free GSH which is responsible for GSH's antioxidant activity. This decrease correlated with an increase in the growth of Mycobacterium tuberculosis (M. tb) in macrophages from HIV(+) individuals. In addition, we observed increased levels of free radicals, interleukin-1 (IL-1), interleukin-17 (IL-17) and transforming growth factor-β (TGF-β) in plasma samples derived from HIV(+) individuals compared to healthy subjects. We observed decreased expression of the genes coding for enzymes responsible for de novo synthesis of GSH in macrophages derived from HIV(+) subjects using quantitative PCR (qPCR). Our results indicate that overproduction of proinflammatory cytokines in HIV(+) individuals lead to increased production of free radicals. This combined with the decreased expression of GSH synthesis enzymes leads to a depletion of free GSH and may lead in part to the loss of immune function observed in HIV patients.
Figures

References
-
- World Health Organization. Global summary of the AIDS epidemic. 2009. http://www.who.int/hiv/data/2009_global_summary.png.
-
- Chiang CY, Centis R, Migliori GB. Drug-resistant tuberculosis: past, present, future. Respirology. 2010;15(3):413–432. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

