Mechanism of FGF23 processing in fibrous dysplasia
- PMID: 22247037
- PMCID: PMC7448291
- DOI: 10.1002/jbmr.1546
Mechanism of FGF23 processing in fibrous dysplasia
Abstract
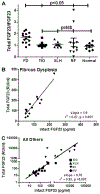
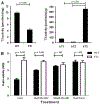
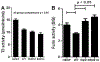
Fibroblast growth factor-23 (FGF23) is a phosphate- and vitamin D-regulating hormone derived from osteoblasts/osteocytes that circulates in both active (intact, iFGF23) and inactive (C-terminal, cFGF23) forms. O-glycosylation by O-glycosyl transferase N-acetylgalactosaminyltransferase 3 (ppGalNAcT3) and differential cleavage by furin have been shown to be involved in regulating the ratio of active to inactive FGF23. Elevated iFGF23 levels are observed in a number of hypophosphatemic disorders, such as X-linked, autosomal recessive, and autosomal dominant hypophosphatemic rickets, whereas low iFGF23 levels are found in the hyperphosphatemic disorder familial tumoral calcinosis/hyperphosphatemic hyperostosis syndrome. Fibrous dysplasia of bone (FD) is associated with increased total FGF23 levels (cFGF23 + iFGF23); however, classic hypophosphatemic rickets is uncommon. Our results suggest that it can be explained by increased FGF23 cleavage leading to an increase in inactive cFGF23 relative to active iFGF23. Given the fact that FD is caused by activating mutations in the small G-protein G(s) α that results in increased cyclic adenosine monophosphate (cAMP) levels, we postulated that there may be altered FGF23 cleavage in FD and that the mechanism may involve alterations in cAMP levels and ppGalNacT3 and furin activities. Analysis of blood specimens from patients with FD confirmed that the elevated total FGF23 levels are the result of proportionally increased cFGF23 levels, consistent with less glycosylation and enhanced cleavage by furin. Analysis of primary cell lines of normal and mutation-harboring bone marrow stromal cells (BMSCs) from patients with FD demonstrated that BMSCs harboring the causative G(s) α mutation had higher cAMP levels, lower ppGalNAcT3, and higher furin activity. These data support the model wherein glycosylation by ppGalNAcT3 inhibits FGF23 cleavage by furin and suggest that FGF23 processing is a regulated process that controls overall FGF23 activity in FD patients.
Copyright © 2012 American Society for Bone and Mineral Research.
Conflict of interest statement
Disclosures
All the authors state that they have no conflicts of interest.
Figures
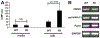

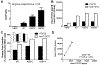
References
-
- White KE, Larsson TE, Econs MJ. The roles of specific genes implicated as circulating factors involved in normal and disordered phosphate homeostasis: frizzled related protein-4, matrix extracellular phosphoglycoprotein, and fibroblast growth factor 23. Endocr Rev. 2006;27:221–41. - PubMed
-
- Prie D, Beck L, Urena P, Friedlander G. Recent findings in phosphate homeostasis. Curr Opin Nephrol Hypertens. 2005;14:318–24. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

