The apoplastic oxidative burst peroxidase in Arabidopsis is a major component of pattern-triggered immunity
- PMID: 22247251
- PMCID: PMC3289579
- DOI: 10.1105/tpc.111.093039
The apoplastic oxidative burst peroxidase in Arabidopsis is a major component of pattern-triggered immunity
Abstract
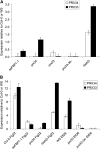
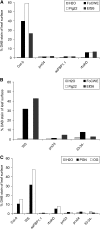
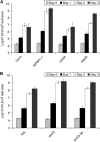
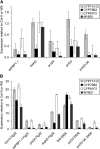
In plants, reactive oxygen species (ROS) associated with the response to pathogen attack are generated by NADPH oxidases or apoplastic peroxidases. Antisense expression of a heterologous French bean (Phaseolus vulgaris) peroxidase (FBP1) cDNA in Arabidopsis thaliana was previously shown to diminish the expression of two Arabidopsis peroxidases (peroxidase 33 [PRX33] and PRX34), block the oxidative burst in response to a fungal elicitor, and cause enhanced susceptibility to a broad range of fungal and bacterial pathogens. Here we show that mature leaves of T-DNA insertion lines with diminished expression of PRX33 and PRX34 exhibit reduced ROS and callose deposition in response to microbe-associated molecular patterns (MAMPs), including the synthetic peptides Flg22 and Elf26 corresponding to bacterial flagellin and elongation factor Tu, respectively. PRX33 and PRX34 knockdown lines also exhibited diminished activation of Flg22-activated genes after Flg22 treatment. These MAMP-activated genes were also downregulated in unchallenged leaves of the peroxidase knockdown lines, suggesting that a low level of apoplastic ROS production may be required to preprime basal resistance. Finally, the PRX33 knockdown line is more susceptible to Pseudomonas syringae than wild-type plants. In aggregate, these data demonstrate that the peroxidase-dependent oxidative burst plays an important role in Arabidopsis basal resistance mediated by the recognition of MAMPs.
Figures


Similar articles
-
A peroxidase-dependent apoplastic oxidative burst in cultured Arabidopsis cells functions in MAMP-elicited defense.Plant Physiol. 2012 Apr;158(4):2013-27. doi: 10.1104/pp.111.190140. Epub 2012 Feb 7. Plant Physiol. 2012. PMID: 22319074 Free PMC article.
-
Apoplastic peroxidases are required for salicylic acid-mediated defense against Pseudomonas syringae.Phytochemistry. 2015 Apr;112:110-21. doi: 10.1016/j.phytochem.2014.07.010. Epub 2014 Aug 2. Phytochemistry. 2015. PMID: 25096754 Free PMC article.
-
The receptor-like cytoplasmic kinase PCRK1 contributes to pattern-triggered immunity against Pseudomonas syringae in Arabidopsis thaliana.New Phytol. 2015 Jul;207(1):78-90. doi: 10.1111/nph.13345. Epub 2015 Feb 25. New Phytol. 2015. PMID: 25711411
-
The apoplastic oxidative burst in response to biotic stress in plants: a three-component system.J Exp Bot. 2002 May;53(372):1367-76. J Exp Bot. 2002. PMID: 11997382 Review.
-
Reactive oxygen species and their role in plant defence and cell wall metabolism.Planta. 2012 Sep;236(3):765-79. doi: 10.1007/s00425-012-1696-9. Epub 2012 Jul 6. Planta. 2012. PMID: 22767200 Review.
Cited by
-
Selective redox signaling shapes plant-pathogen interactions.Plant Physiol. 2021 May 27;186(1):53-65. doi: 10.1093/plphys/kiaa088. Plant Physiol. 2021. PMID: 33793940 Free PMC article. Review.
-
Growth deficiency and enhanced basal immunity in Arabidopsis thaliana mutants of EDM2, EDM3 and IBM2 are genetically interlinked.PLoS One. 2024 Feb 8;19(2):e0291705. doi: 10.1371/journal.pone.0291705. eCollection 2024. PLoS One. 2024. PMID: 38329997 Free PMC article.
-
Analysis of the Oxidative Burst and Its Relevant Signaling Pathways in Leptosphaeria maculans-Brassica napus Pathosystem.Int J Mol Sci. 2021 May 1;22(9):4812. doi: 10.3390/ijms22094812. Int J Mol Sci. 2021. PMID: 34062819 Free PMC article.
-
Comprehensive analysis of the Ppatg3 mutant reveals that autophagy plays important roles in gametophore senescence in Physcomitrella patens.BMC Plant Biol. 2020 Sep 23;20(1):440. doi: 10.1186/s12870-020-02651-6. BMC Plant Biol. 2020. PMID: 32967624 Free PMC article.
-
Heterologous Expression of Human Metallothionein Gene HsMT1L Can Enhance the Tolerance of Tobacco (Nicotiana nudicaulis Watson) to Zinc and Cadmium.Genes (Basel). 2022 Dec 19;13(12):2413. doi: 10.3390/genes13122413. Genes (Basel). 2022. PMID: 36553680 Free PMC article.
References
-
- Ausubel F.M., Brent R., Kingston R.E., Moore D.D., Seidman J.G., Smith J.A., Struhl K. (1994). Current Protocols in Molecular Biology. (New York: John Wiley & Sons; ).
-
- Bolwell G.P. (1999). Role of active oxygen species and NO in plant defence responses. Curr. Opin. Plant Biol. 2: 287–294 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

