Sgt1 acts via an LKB1/AMPK pathway to establish cortical polarity in larval neuroblasts
- PMID: 22248825
- PMCID: PMC4013783
- DOI: 10.1016/j.ydbio.2011.12.047
Sgt1 acts via an LKB1/AMPK pathway to establish cortical polarity in larval neuroblasts
Abstract
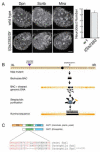
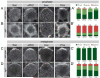
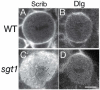
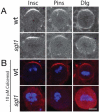
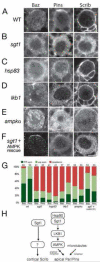
Drosophila neuroblasts are a model system for studying stem cell self-renewal and the establishment of cortical polarity. Larval neuroblasts generate a large apical self-renewing neuroblast, and a small basal cell that differentiates. We performed a genetic screen to identify regulators of neuroblast self-renewal, and identified a mutation in sgt1 (suppressor-of-G2-allele-of-skp1) that had fewer neuroblasts. We found that sgt1 neuroblasts have two polarity phenotypes: failure to establish apical cortical polarity at prophase, and lack of cortical Scribble localization throughout the cell cycle. Apical cortical polarity was partially restored at metaphase by a microtubule-induced cortical polarity pathway. Double mutants lacking Sgt1 and Pins (a microtubule-induced polarity pathway component) resulted in neuroblasts without detectable cortical polarity and formation of "neuroblast tumors." Mutants in hsp83 (encoding the predicted Sgt1-binding protein Hsp90), LKB1, or AMPKα all show similar prophase apical cortical polarity defects (but no Scribble phenotype), and activated AMPKα rescued the sgt1 mutant phenotype. We propose that an Sgt1/Hsp90-LKB1-AMPK pathway acts redundantly with a microtubule-induced polarity pathway to generate neuroblast cortical polarity, and the absence of neuroblast cortical polarity can produce neuroblast tumors.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Lgl, Pins and aPKC regulate neuroblast self-renewal versus differentiation.Nature. 2006 Feb 2;439(7076):594-8. doi: 10.1038/nature04299. Epub 2005 Dec 14. Nature. 2006. PMID: 16357871
-
Microtubule-induced Pins/Galphai cortical polarity in Drosophila neuroblasts.Cell. 2005 Dec 29;123(7):1323-35. doi: 10.1016/j.cell.2005.09.043. Cell. 2005. PMID: 16377571
-
Drosophila Aurora-A kinase inhibits neuroblast self-renewal by regulating aPKC/Numb cortical polarity and spindle orientation.Genes Dev. 2006 Dec 15;20(24):3464-74. doi: 10.1101/gad.1489406. Genes Dev. 2006. PMID: 17182871 Free PMC article.
-
The Case of the Scribble Polarity Module in Asymmetric Neuroblast Division in Development and Tumorigenesis.Int J Mol Sci. 2020 Apr 20;21(8):2865. doi: 10.3390/ijms21082865. Int J Mol Sci. 2020. PMID: 32325951 Free PMC article. Review.
-
Mechanisms of asymmetric progenitor divisions in the Drosophila central nervous system.Adv Exp Med Biol. 2013;786:79-102. doi: 10.1007/978-94-007-6621-1_6. Adv Exp Med Biol. 2013. PMID: 23696353 Review.
Cited by
-
Identification of protein quality control regulators using a Drosophila model of TPI deficiency.Neurobiol Dis. 2021 May;152:105299. doi: 10.1016/j.nbd.2021.105299. Epub 2021 Feb 15. Neurobiol Dis. 2021. PMID: 33600953 Free PMC article.
-
SUGT1 regulates the progression of ovarian cancer through the AKT/PI3K/mTOR signaling pathway.Transl Oncol. 2024 Nov;49:102088. doi: 10.1016/j.tranon.2024.102088. Epub 2024 Aug 20. Transl Oncol. 2024. PMID: 39167956 Free PMC article.
-
Scrib regulates HGF-mediated epithelial morphogenesis and is stabilized by Sgt1-HSP90.J Cell Sci. 2012 Sep 1;125(Pt 17):4147-57. doi: 10.1242/jcs.108670. Epub 2012 May 23. J Cell Sci. 2012. PMID: 22623728 Free PMC article.
-
The Role of AMPK in Drosophila melanogaster.Exp Suppl. 2016;107:389-401. doi: 10.1007/978-3-319-43589-3_16. Exp Suppl. 2016. PMID: 27812989 Free PMC article. Review.
-
LKB1 deletion causes early changes in atrial channel expression and electrophysiology prior to atrial fibrillation.Cardiovasc Res. 2015 Oct 1;108(1):197-208. doi: 10.1093/cvr/cvv212. Cardiovasc Res. 2015. PMID: 26378152 Free PMC article.
References
-
- Ahmed A, Chandra S, Magarinos M, Vaessin H. Echinoid mutants exhibit neurogenic phenotypes and show synergistic interactions with the Notch signaling pathway. Development. 2003;130:6295–6304. - PubMed
-
- Barros CS, Phelps CB, Brand AH. Drosophila nonmuscle myosin II promotes the asymmetric segregation of cell fate determinants by cortical exclusion rather than active transport. Dev Cell. 2003;5:829–840. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

