A molecular switch on an arrestin-like protein relays glucose signaling to transporter endocytosis
- PMID: 22249293
- PMCID: PMC3265958
- DOI: 10.1083/jcb.201109113
A molecular switch on an arrestin-like protein relays glucose signaling to transporter endocytosis
Abstract
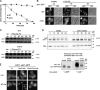
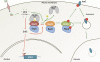
Endocytosis regulates the plasma membrane protein landscape in response to environmental cues. In yeast, the endocytosis of transporters depends on their ubiquitylation by the Nedd4-like ubiquitin ligase Rsp5, but how extracellular signals trigger this ubiquitylation is unknown. Various carbon source transporters are known to be ubiquitylated and endocytosed when glucose-starved cells are exposed to glucose. We show that this required the conserved arrestin-related protein Rod1/Art4, which was activated in response to glucose addition. Indeed, Rod1 was a direct target of the glucose signaling pathway composed of the AMPK homologue Snf1 and the PP1 phosphatase Glc7/Reg1. Glucose promoted Rod1 dephosphorylation and its subsequent release from a phospho-dependent interaction with 14-3-3 proteins. Consequently, this allowed Rod1 ubiquitylation by Rsp5, which was a prerequisite for transporter endocytosis. This paper therefore demonstrates that the arrestin-related protein Rod1 relays glucose signaling to transporter endocytosis and provides the first molecular insights into the nutrient-induced activation of an arrestin-related protein through a switch in post-translational modifications.
Figures



References
-
- Bhalla V., Daidié D., Li H., Pao A.C., LaGrange L.P., Wang J., Vandewalle A., Stockand J.D., Staub O., Pearce D. 2005. Serum- and glucocorticoid-regulated kinase 1 regulates ubiquitin ligase neural precursor cell-expressed, developmentally down-regulated protein 4-2 by inducing interaction with 14-3-3. Mol. Endocrinol. 19:3073–3084 10.1210/me.2005-0193 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

