Individual differences in neurocognitive aging of the medial temporal lobe
- PMID: 22253491
- PMCID: PMC3259151
- DOI: 10.1007/s11357-006-9017-5
Individual differences in neurocognitive aging of the medial temporal lobe
Abstract
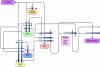
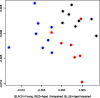
A wide spectrum of outcomes in the cognitive effects of aging is routinely observed in studies of the elderly. Individual differences in neurocognitive aging are also a characteristic of other species, such as rodents and non-human primates. In particular, investigations at behavioral, brain systems, cellular and molecular levels of analysis have provided much information on the basis for individual differences in neurocognitive aging among healthy outbred rats. These findings are likely to be relevant to an understanding of the effects of aging on the brain, apart from neurodegenerative conditions, such as Alzheimer's disease, which do not naturally occur in rodents. Here we review and integrate those findings in a model supporting the concept that certain features of cognitive decline are caused by distributed alterations in the medial temporal lobe, which alter the information processing functions of the hippocampal formation. An additional emerging concept from this research is that preserved abilities at older ages may depend on adaptive changes in the hippocampal system that distinguish successful aging.
Keywords: cognitive impairment; hippocampal formation; rats; spatial memory; successful aging.
Figures
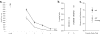


References
LinkOut - more resources
Full Text Sources
