Dietary advanced glycation end products and aging
- PMID: 22254007
- PMCID: PMC3257625
- DOI: 10.3390/nu2121247
Dietary advanced glycation end products and aging
Abstract
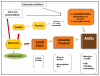
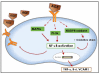
Advanced glycation end products (AGEs) are a heterogeneous, complex group of compounds that are formed when reducing sugar reacts in a non-enzymatic way with amino acids in proteins and other macromolecules. This occurs both exogenously (in food) and endogenously (in humans) with greater concentrations found in older adults. While higher AGEs occur in both healthy older adults and those with chronic diseases, research is progressing to both quantify AGEs in food and in people, and to identify mechanisms that would explain why some human tissues are damaged, and others are not. In the last twenty years, there has been increased evidence that AGEs could be implicated in the development of chronic degenerative diseases of aging, such as cardiovascular disease, Alzheimer's disease and with complications of diabetes mellitus. Results of several studies in animal models and humans show that the restriction of dietary AGEs has positive effects on wound healing, insulin resistance and cardiovascular diseases. Recently, the effect of restriction in AGEs intake has been reported to increase the lifespan in animal models. This paper will summarize the work that has been published for both food AGEs and in vivo AGEs and their relation with aging, as well as provide suggestions for future research.
Keywords: Maillard reaction; advanced glycation end products; aging.
Figures
References
-
- Brownlee M., Vlassara H., Cerami A. Nonenzimatic glycosilation and the pathogenesis of diabetes complications. Ann. Intern. Med. 1984;101:527–537. - PubMed
-
- Ahmed N. Advanced glycation endproducts—role in pathology of diabetic complications. Diabetes Res. Clin. Pract. 2005;67:3–21. - PubMed
-
- Uribarri J., Tuttle K.R. Advanced glycation end products and nephrotoxicity of high-protein diets. Clin. J. Am. Soc. Nephrol. 2006;1:1293–1299. - PubMed
-
- Ahmed N., Thornalley P.J. Quantitative screening of protein biomarkers of early glycation, advanced glycation, oxidation and nitrosation in cellular and extracellular proteins by tandem mass spectrometry multiple reaction monitoring. Biochem. Soc. Trans. 2003;31:1417–1422. doi: 10.1042/BST0311417. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical