Potential health-modulating effects of isoflavones and metabolites via activation of PPAR and AhR
- PMID: 22254019
- PMCID: PMC3257647
- DOI: 10.3390/nu2030241
Potential health-modulating effects of isoflavones and metabolites via activation of PPAR and AhR
Abstract
Isoflavones have multiple actions on cell functions. The most prominent one is the activation of estrogen receptors. Other functions are often overlooked, but are equally important and explain the beneficial health effects of isoflavones. Isoflavones are potent dual PPARα/γ agonists and exert anti-inflammatory activity, which may contribute to the prevention of metabolic syndrome, atherosclerosis and various other inflammatory diseases. Some isoflavones are potent aryl hydrocarbon receptor (AhR) agonists and induce cell cycle arrest, chemoprevention and modulate xenobiotic metabolism. This review discusses effects mediated by the activation of AhR and PPARs and casts a light on the concerted action of isoflavones.
Keywords: AhR; PPARα; PPARγ; atherosclerosis; cell cycle control; inflammation; isoflavones; metabolic syndrome; xenobiotic metabolism.
Figures
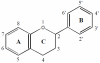
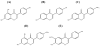
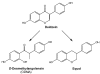
Similar articles
-
Red clover isoflavones biochanin A and formononetin are potent ligands of the human aryl hydrocarbon receptor.J Steroid Biochem Mol Biol. 2008 Jan;108(1-2):171-7. doi: 10.1016/j.jsbmb.2007.10.001. Epub 2007 Oct 22. J Steroid Biochem Mol Biol. 2008. PMID: 18060767
-
Effects of the environmental contaminants DEHP and TCDD on estradiol synthesis and aryl hydrocarbon receptor and peroxisome proliferator-activated receptor signalling in the human granulosa cell line KGN.Mol Hum Reprod. 2014 Sep;20(9):919-28. doi: 10.1093/molehr/gau045. Epub 2014 Jun 20. Mol Hum Reprod. 2014. PMID: 24950685
-
Nuclear Receptors in Myocardial and Cerebral Ischemia-Mechanisms of Action and Therapeutic Strategies.Int J Mol Sci. 2021 Nov 15;22(22):12326. doi: 10.3390/ijms222212326. Int J Mol Sci. 2021. PMID: 34830207 Free PMC article. Review.
-
Aryl hydrocarbon receptor (AhR) regulates adipocyte differentiation by assembling CRL4B ubiquitin ligase to target PPARγ for proteasomal degradation.J Biol Chem. 2019 Nov 29;294(48):18504-18515. doi: 10.1074/jbc.RA119.009282. Epub 2019 Oct 25. J Biol Chem. 2019. PMID: 31653699 Free PMC article.
-
Peroxisome proliferator-activated receptors (PPARs) and the human skin: importance of PPARs in skin physiology and dermatologic diseases.Am J Clin Dermatol. 2008;9(1):15-31. doi: 10.2165/00128071-200809010-00002. Am J Clin Dermatol. 2008. PMID: 18092840 Review.
Cited by
-
Polyphenols and Tryptophan Metabolites Activate the Aryl Hydrocarbon Receptor in an in vitro Model of Colonic Fermentation.Mol Nutr Food Res. 2019 Feb;63(3):e1800722. doi: 10.1002/mnfr.201800722. Epub 2018 Nov 28. Mol Nutr Food Res. 2019. PMID: 30443985 Free PMC article.
-
4',6-Dimethoxyisoflavone-7-O-β-D-glucopyranoside (wistin) is a peroxisome proliferator-activated receptor α (PPARα) agonist in mouse hepatocytes.Mol Cell Biochem. 2018 Sep;446(1-2):35-41. doi: 10.1007/s11010-018-3270-7. Epub 2018 Jan 9. Mol Cell Biochem. 2018. PMID: 29318455
-
Isoflavones enhance interleukin-17 gene expression via retinoic acid receptor-related orphan receptors α and γ.Toxicology. 2015 Mar 2;329:32-9. doi: 10.1016/j.tox.2015.01.007. Epub 2015 Jan 9. Toxicology. 2015. PMID: 25583575 Free PMC article.
-
The associations between plasma phytoestrogens concentration and metabolic syndrome risks in Chinese population.PLoS One. 2018 Mar 20;13(3):e0194639. doi: 10.1371/journal.pone.0194639. eCollection 2018. PLoS One. 2018. PMID: 29558501 Free PMC article.
-
Moderate Increase of Indoxyl Sulfate Promotes Monocyte Transition into Profibrotic Macrophages.PLoS One. 2016 Feb 29;11(2):e0149276. doi: 10.1371/journal.pone.0149276. eCollection 2016. PLoS One. 2016. PMID: 26925780 Free PMC article.
References
-
- Haslam E. Practical Polyphenolics: from Structure to Molecular Recognition and Physiological Action. XV. Cambridge Univ. Press; Cambridge, UK: 1998. p. 422.
-
- Di Carlo G., Mascolo N., Izzo A.A., Capasso F. Flavonoids: old and new aspects of a class of natural therapeutic drugs. Life Sci. 1999;65:337–353. - PubMed
-
- Peterson J., Dwyer J. Flavonoids: Dietary occurrence and biochemical activity. Nutrition Res. 1998;18:1995–2018.
-
- Mazur W., Adlercreutz H. Overview of naturally occurring endocrine-active substances in the human diet in relation to human health. Nutrition. 2000;16:654–658. - PubMed
-
- Liggins J., Bluck L.J., Runswick S., Atkinson C., Coward W.A., Bingham S.A. Daidzein and genistein content of fruits and nuts. J. Nutr. Biochem. 2000;11:326–331. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

