A randomized, double-blind, placebo-controlled study of the safety and tolerance of regadenoson in subjects with stage 3 or 4 chronic kidney disease
- PMID: 22259009
- PMCID: PMC3332362
- DOI: 10.1007/s12350-011-9508-3
A randomized, double-blind, placebo-controlled study of the safety and tolerance of regadenoson in subjects with stage 3 or 4 chronic kidney disease
Abstract
Background: The safety and tolerability of regadenoson, a pharmacologic stress agent that is excreted primarily by the kidneys, were examined in subjects with chronic kidney disease (CKD).
Methods: This multicenter, double-blind, randomized, placebo-controlled study involved men and women, ≥18 years of age, with stage 3 or 4 [estimated glomerular filtration rate (eGFR) 30-59 mL/minute/1.73 m(2) and 15-29 mL/minute/1.73 m(2), respectively] CKD and known or suspected coronary artery disease. Subjects were randomized 2:1 to receive one 10-second intravenous injection of regadenoson 0.4 mg or placebo. The primary outcome measure was the frequency of serious adverse events over 24-h post-dose.
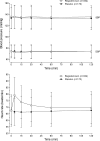
Results: The study included 432 subjects with stage 3 (regadenoson n = 287; placebo n = 145) and 72 with stage 4 (regadenoson n = 47; placebo n = 25) CKD. No serious adverse events or deaths were reported over 24-h post-dose. The overall adverse event incidence was higher with regadenoson than placebo (62.6% vs 21.2%; P < .0001). Of the most common adverse events (≥5%) reported by subjects receiving regadenoson, headache (24.9% vs 7.1%), dyspnea (19.2% vs 0.6%), chest discomfort (14.7% vs 0.6%), nausea (14.7% vs 1.2%), flushing (12.0% vs 1.8%), and dizziness (9.6% vs 0.6%) occurred significantly more often (P < .0001) with regadenoson than placebo. There were no trends for clinically meaningful changes in eGFR from baseline to 24-h post-dose in subjects with stage 3 or 4 CKD.
Conclusions: Regadenoson was not associated with any serious or unexpected adverse events in subjects with stage 3 or 4 CKD.
Figures
References
-
- Gao Z, Li Z, Baker SP, Lasley RD, Meyer S, Elzein E, et al. Novel short-acting A2A adenosine receptor agonists for coronary vasodilation: Inverse relationship between affinity and duration of action of A2A agonists. J Pharmacol Exp Ther. 2001;298:209–218. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous