STAT1-deficient mice spontaneously develop estrogen receptor α-positive luminal mammary carcinomas
- PMID: 22264274
- PMCID: PMC3496133
- DOI: 10.1186/bcr3100
STAT1-deficient mice spontaneously develop estrogen receptor α-positive luminal mammary carcinomas
Abstract
Introduction: Although breast cancers expressing estrogen receptor-α (ERα) and progesterone receptors (PR) are the most common form of mammary malignancy in humans, it has been difficult to develop a suitable mouse model showing similar steroid hormone responsiveness. STAT transcription factors play critical roles in mammary gland tumorigenesis, but the precise role of STAT1 remains unclear. Herein, we show that a subset of human breast cancers display reduced STAT1 expression and that mice lacking STAT1 surprisingly develop ERα+/PR+ mammary tumors.
Methods: We used a combination of approaches, including histological examination, gene targeted mice, gene expression analysis, tumor transplantaion, and immunophenotyping, to pursue this study.
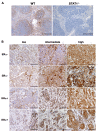
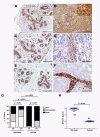
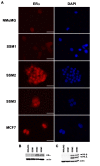
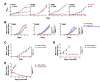
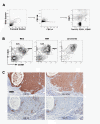
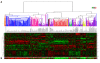
Results: Forty-five percent (37/83) of human ERα+ and 22% (17/78) of ERα- breast cancers display undetectable or low levels of STAT1 expression in neoplastic cells. In contrast, STAT1 expression is elevated in epithelial cells of normal breast tissues adjacent to the malignant lesions, suggesting that STAT1 is selectively downregulated in the tumor cells during tumor progression. Interestingly, the expression levels of STAT1 in the tumor-infiltrating stromal cells remain elevated, indicating that single-cell resolution analysis of STAT1 level in primary breast cancer biopsies is necessary for accurate assessment. Female mice lacking functional STAT1 spontaneously develop mammary adenocarcinomas that comprise > 90% ERα+/PR+ tumor cells, and depend on estrogen for tumor engraftment and progression. Phenotypic marker analyses demonstrate that STAT1-/- mammary tumors arise from luminal epithelial cells, but not myoepithelial cells. In addition, the molecular signature of the STAT1-/- mammary tumors overlaps closely to that of human luminal breast cancers. Finally, introduction of wildtype STAT1, but not a STAT1 mutant lacking the critical Tyr701 residue, into STAT1-/- mammary tumor cells results in apoptosis, demonstrating that the tumor suppressor function of STAT1 is cell-autonomous and requires its transcriptional activity.
Conclusions: Our findings demonstrate that STAT1 suppresses mammary tumor formation and its expression is frequently lost during breast cancer progression. Spontaneous mammary tumors that develop in STAT1-/- mice closely recapitulate the progression, ovarian hormone responsiveness, and molecular characteristics of human luminal breast cancer, the most common subtype of human breast neoplasms, and thus represent a valuable platform for testing novel treatments and detection modalities.
Figures
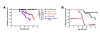

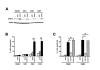
References
-
- Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lonning PE, Borresen-Dale AL, Brown PO, Botstein D. Molecular portraits of human breast tumours. Nature. 2000;406:747–752. doi: 10.1038/35021093. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

