HSF-1 regulators DDL-1/2 link insulin-like signaling to heat-shock responses and modulation of longevity
- PMID: 22265419
- PMCID: PMC3615449
- DOI: 10.1016/j.cell.2011.12.019
HSF-1 regulators DDL-1/2 link insulin-like signaling to heat-shock responses and modulation of longevity
Abstract
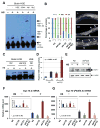
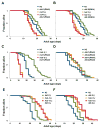
Extended longevity is often correlated with increased resistance against various stressors. Insulin/IGF-1-like signaling (IIS) is known to have a conserved role in aging and cellular mechanisms against stress. In C. elegans, genetic studies suggest that heat-shock transcription factor HSF-1 is required for IIS to modulate longevity. Here, we report that the activity of HSF-1 is regulated by IIS. This regulation occurs at an early step of HSF-1 activation via two HSF-1 regulators, DDL-1 and DDL-2. Inhibition of DDL-1/2 increases longevity and thermotolerance in an hsf-1-dependent manner. Furthermore, biochemical analyses suggest that DDL-1/2 negatively regulate HSF-1 activity by forming a protein complex with HSF-1. The formation of this complex (DHIC) is affected by the phosphorylation status of DDL-1. Both the formation of DHIC and the phosphorylation of DDL-1 are controlled by IIS. Our findings point to DDL-1/2 as a link between IIS and the HSF-1 pathway.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




References
-
- Clos J, Westwood JT, Becker PB, Wilson S, Lambert K, Wu C. Molecular cloning and expression of a hexameric Drosophila heat shock factor subject to negative regulation. Cell. 1990;63:1085–1097. - PubMed
-
- Cohen E, Bieschke J, Perciavalle RM, Kelly JW, Dillin A. Opposing activities protect against age-onset proteotoxicity. Science. 2006;313:1604–1610. - PubMed
-
- Derivery E, Sousa C, Gautier JJ, Lombard B, Loew D, Gautreau A. The Arp2/3 activator WASH controls the fission of endosomes through a large multiprotein complex. Developmental cell. 2009;17:712–723. - PubMed
-
- Finkel T, Holbrook NJ. Oxidants, oxidative stress and the biology of ageing. Nature. 2000;408:239–247. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

