Oral repeated-dose toxicity studies of coenzyme Q10 in beagle dogs
- PMID: 22267890
- PMCID: PMC3288259
- DOI: 10.1177/1091581811425256
Oral repeated-dose toxicity studies of coenzyme Q10 in beagle dogs
Abstract
To support phase III testing of coenzyme Q10 (CoQ₁₀) in humans, we conducted pharmacokinetic and toxicology studies in beagle dogs. Following single gavage administration of CoQ₁₀ at 600, 1200, 1800, or 2400 mg/kg per d no obvious dose response was observed in maximum concentration (C(max)) or area under the curve (AUC) versus time curve at the 3 highest dosages. In a repeated-dose study of CoQ₁₀ at 600, 1200, 1800, or 2400 mg/kg per d for 4 weeks, CoQ₁₀ reached steady state in plasma by 2 weeks at all dosages. Both C (max) and AUC increased with increasing dosage of CoQ₁₀. The highest plasma levels were recorded at 1800 mg/kg per d. In a 39-week chronic toxicity study of CoQ₁₀ at 1200 and 1800 mg/kg per d or placebo, CoQ₁₀ reached steady state in plasma by 13 weeks. Behaviors, blood chemistries, and detailed histopathology were normal. No deaths occurred. These results support the use of a 2400 mg/d dosage of CoQ₁₀ in human clinical trials.
Figures

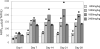



References
-
- [accessed July 2011]; ClinicalTrials.gov Identifiers NCT00608881 and NCT00740714.
-
- Lehninger AL. The molecular organization of mitochondrial membranes. Adv Cytopharmacol. 1971;1:199–208. - PubMed
-
- Beal MF. Mitochondria, oxidative damage, and inflammation in Parkinson's disease. Ann N Y Acad Sci. 2003;991:120–131. - PubMed
-
- Schilling G, Coonfield ML, Ross CA, et al. Coenzyme Q10 and remacemide hydrochloride ameliorate motor deficits in a Huntington's disease transgenic mouse model. Neurosci Lett. 2001;315(3):149–153. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

