Renal function in HIV-infected children and adolescents treated with tenofovir disoproxil fumarate and protease inhibitors
- PMID: 22269183
- PMCID: PMC3306735
- DOI: 10.1186/1471-2334-12-18
Renal function in HIV-infected children and adolescents treated with tenofovir disoproxil fumarate and protease inhibitors
Abstract
Background: Kidney disease is an important complication in HIV infected people, and this may be related to infection or antiretroviral therapy (ART). Our aim is to assess renal function in HIV infected paediatric patients, who may be particularly affected and are likely to take ART for longer than adults, and investigate the long term role of Tenofovir Disoproxil Fumarate (TDF) alone or co-administered with Ritonavir-boosted Protease Inhibitors (PI).
Methods: Serum creatinine, phosphate and potassium levels, with estimated Glomerular Filtration Rate (eGFR), had been prospectively evaluated for 2 years in a cohort of HIV infected children and adolescents (age 9-18) on ART, and data analyzed according to the exposure to TDF or simultaneous TDF and PI.
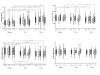
Results: Forty-nine patients were studied (57% female, mean age 14). Sixty-three percent were treated with ART containing TDF (Group A), and 37% without TDF (Group B); 47% with concomitant use of TDF and PI (Group C) and 53% without this combination (Group D). The groups didn't differ for age, gender or ethnicity. The median creatinine increased in the entire cohort and in all the groups analyzed; eGFR decreased from 143.6 mL/min/1.73 m2 at baseline to 128.9 after 2 years (p = 0.006) in the entire cohort. Three patients presented a mild eGFR reduction, all were on TDF+PI. Phosphatemia decreased significantly in the entire cohort (p = 0.0003) and in TDF+PI group (p = 0.0128) after 2 years. Five patients (10%) developed hypophosphatemia (Division of Acquired Immune Deficiency AE grade 1 or 2), and four of them were on TDF+PI.
Conclusions: Renal function decrease and hypophosphatemia occur over time in HIV infected children and adolescents on ART. The association with co-administration of TDF and PI appears weak, and further studies are warranted.
Figures
References
-
- D'Arminio Monforte A, Sabin CA, Phillips A. et al. The changing incidence of AIDS events in patients receiving highly active antiretroviral therapy. Arc Intern Med. 2005;165:416–423. - PubMed
-
- Guideline on the clinical development of medical products for the treatment of HIV infection. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guidelin...
-
- Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Department of Health and Human Services. 2011. pp. 1–166. Available at [ http://www.aidsinfo.nih.gov/ContentFiles/AdultandAdolescentGL.pdf]
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

