Flp and Cre expressed from Flp-2A-Cre and Flp-IRES-Cre transcription units mediate the highest level of dual recombinase-mediated cassette exchange
- PMID: 22270085
- PMCID: PMC3333864
- DOI: 10.1093/nar/gks027
Flp and Cre expressed from Flp-2A-Cre and Flp-IRES-Cre transcription units mediate the highest level of dual recombinase-mediated cassette exchange
Abstract
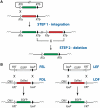
Recombinase-mediated cassette exchange (RMCE) is a powerful tool for unidirectional integration of DNA fragments of interest into a pre-determined genome locale. In this report, we examined how the efficiency of dual RMCE catalyzed by Flp and Cre depends on the nature of transcription units that express the recombinases. The following recombinase transcription units were analyzed: (i) Flp and Cre genes expressed as individual transcription units located on different vectors, (ii) Flp and Cre genes expressed as individual transcription units located on the same vector, (iii) Flp and Cre genes expressed from a single promoter and separated by internal ribosome entry sequence and (iv) Flp and Cre coding sequences separated by the 2A peptide and expressed as a single gene. We found that the highest level of dual RMCE (35-45% of the transfected cells) can be achieved when Flp and Cre recombinases are expressed as Flp-2A-Cre and Flp-IRES-Cre transcription units. In contrast, the lowest level of dual RMCE (∼1% of the transfected cells) is achieved when Flp and Cre are expressed as individual transcription units. The analysis shows that it is the relative Flp-to-Cre ratio that critically affects the efficiency of dual RMCE. Our results will be helpful for maximizing the efficiency of dual RMCE aimed to engineer and re-engineer genomes.
Figures


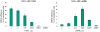

References
-
- Schlake T, Bode J. Use of mutated FLP recognition target (FRT) sites for the exchange of expression cassettes at defined chromosomal loci. Biochemistry. 1994;33:12746–12751. - PubMed
-
- Belteki G, Gertsenstein M, Ow DW, Nagy A. Site-specific cassette exchange and germline transmission with mouse ES cells expressing phiC31 integrase. Nat. Biotechnol. 2003;21:321–324. - PubMed
-
- Glaser S, Anastassiadis K, Stewart AF. Current issues in mouse genome engineering. Nat. Genet. 2005;37:1187–1193. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

