JAM-A protects from thrombosis by suppressing integrin αIIbβ3-dependent outside-in signaling in platelets
- PMID: 22271446
- PMCID: PMC3321861
- DOI: 10.1182/blood-2011-12-397398
JAM-A protects from thrombosis by suppressing integrin αIIbβ3-dependent outside-in signaling in platelets
Abstract
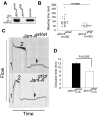
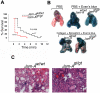
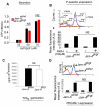
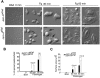
Mounting evidence suggests that agonist-initiated signaling in platelets is closely regulated to avoid excessive responses to injury. A variety of physiologic agonists induce a cascade of signaling events termed as inside-out signaling that culminate in exposure of high-affinity binding sites on integrin α(IIb)β(3). Once platelet activation has occurred, integrin α(IIb)β(3) stabilizes thrombus formation by providing agonist-independent "outside-in" signals mediated in part by contractile signaling. Junctional adhesion molecule A (JAM-A), a member of the cortical thymocyte marker of the Xenopus (CTX) family, was initially identified as a receptor for a platelet stimulatory mAb. Here we show that JAM-A in resting platelets functions as an endogenous inhibitor of platelet function. Genetic ablation of Jam-A in mice enhances thrombotic function of platelets in vivo. The absence of Jam-A results in increase in platelet aggregation ex vivo. This gain of function is not because of enhanced inside-out signaling because granular secretion, Thromboxane A2 (TxA2) generation, as well as fibrinogen receptor activation, are normal in the absence of Jam-A. Interestingly, integrin outside-in signaling such as platelet spreading and clot retraction is augmented in Jam-A-deficient platelets. We conclude that JAM-A normally limits platelet accumulation by inhibiting integrin outside-in signaling thus preventing premature platelet activation.
Figures



Comment in
-
JAMming the signals.Blood. 2012 Apr 5;119(14):3197-8. doi: 10.1182/blood-2012-02-409060. Blood. 2012. PMID: 22493215
Similar articles
-
Junctional adhesion molecule-A suppresses platelet integrin αIIbβ3 signaling by recruiting Csk to the integrin-c-Src complex.Blood. 2014 Feb 27;123(9):1393-402. doi: 10.1182/blood-2013-04-496232. Epub 2013 Dec 3. Blood. 2014. PMID: 24300854 Free PMC article.
-
Hyperreactivity of junctional adhesion molecule A-deficient platelets accelerates atherosclerosis in hyperlipidemic mice.Circ Res. 2015 Feb 13;116(4):587-99. doi: 10.1161/CIRCRESAHA.116.304035. Epub 2014 Dec 3. Circ Res. 2015. PMID: 25472975
-
Distinct roles for Rap1b protein in platelet secretion and integrin αIIbβ3 outside-in signaling.J Biol Chem. 2011 Nov 11;286(45):39466-77. doi: 10.1074/jbc.M111.239608. Epub 2011 Sep 22. J Biol Chem. 2011. PMID: 21940635 Free PMC article.
-
Integrin αIIbβ3 outside-in signaling.Blood. 2017 Oct 5;130(14):1607-1619. doi: 10.1182/blood-2017-03-773614. Epub 2017 Aug 9. Blood. 2017. PMID: 28794070 Free PMC article. Review.
-
Platelet integrin αIIbβ3: signal transduction, regulation, and its therapeutic targeting.J Hematol Oncol. 2019 Mar 7;12(1):26. doi: 10.1186/s13045-019-0709-6. J Hematol Oncol. 2019. PMID: 30845955 Free PMC article. Review.
Cited by
-
Junctional Adhesion Molecules: Potential Proteins in Atherosclerosis.Front Cardiovasc Med. 2022 Jul 7;9:888818. doi: 10.3389/fcvm.2022.888818. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 35872908 Free PMC article. Review.
-
Junctional adhesion molecule-A: functional diversity through molecular promiscuity.Cell Mol Life Sci. 2018 Apr;75(8):1393-1409. doi: 10.1007/s00018-017-2729-0. Epub 2017 Dec 14. Cell Mol Life Sci. 2018. PMID: 29238845 Free PMC article. Review.
-
Platelet signaling: a complex interplay between inhibitory and activatory networks.J Thromb Haemost. 2016 May;14(5):918-30. doi: 10.1111/jth.13302. Epub 2016 Apr 9. J Thromb Haemost. 2016. PMID: 26929147 Free PMC article. Review.
-
Harnessing the platelet signaling network to produce an optimal hemostatic response.Hematol Oncol Clin North Am. 2013 Jun;27(3):381-409. doi: 10.1016/j.hoc.2013.02.002. Epub 2013 Apr 11. Hematol Oncol Clin North Am. 2013. PMID: 23714305 Free PMC article. Review.
-
Apolipoprotein A-I, elevated in trauma patients, inhibits platelet activation and decreases clot strength.Platelets. 2022 Nov 17;33(8):1119-1131. doi: 10.1080/09537104.2022.2078488. Epub 2022 Jun 5. Platelets. 2022. PMID: 35659185 Free PMC article.
References
-
- Broos K, Feys HB, De Meyer SF, Vanhoorelbeke K, Deckmyn H. Platelets at work in primary hemostasis. Blood Rev. 2011;25(4):155–167. - PubMed
-
- Hughes PE, Pfaff M. Integrin affinity modulation. Trends Cell Biol. 1998;8(9):359–364. - PubMed
-
- Shattil SJ. Signaling through platelet integrin alpha IIb beta 3: inside-out, outside-in, and sideways. Thromb Haemost. 1999;82(2):318–325. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HL63960/HL/NHLBI NIH HHS/United States
- R01 HL119374/HL/NHLBI NIH HHS/United States
- T32 HL007439/HL/NHLBI NIH HHS/United States
- R29 HL057630/HL/NHLBI NIH HHS/United States
- R01 HL063960/HL/NHLBI NIH HHS/United States
- 2P20 16472-11/PHS HHS/United States
- R01 HL057630/HL/NHLBI NIH HHS/United States
- HL57630/HL/NHLBI NIH HHS/United States
- HL40387/HL/NHLBI NIH HHS/United States
- P01 HL040387/HL/NHLBI NIH HHS/United States
- P20 RR015588/RR/NCRR NIH HHS/United States
- P20 RR016472/RR/NCRR NIH HHS/United States
- 5P20 RR015588-10/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

