Risk of advanced fibrosis with grafts from hepatitis C antibody-positive donors: a multicenter cohort study
- PMID: 22271671
- PMCID: PMC3334393
- DOI: 10.1002/lt.23396
Risk of advanced fibrosis with grafts from hepatitis C antibody-positive donors: a multicenter cohort study
Abstract
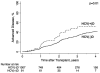
Over the last decade, the use of liver grafts from hepatitis C virus antibody-positive donors [HCV(+)Ds] has tripled in the United States. Although previous studies have demonstrated no association between an HCV(+)D status and graft loss, the effects of an HCV(+)D on histological outcomes are not well known. Hepatitis C virus (HCV)-infected recipients at 5 US centers (2002-2007) who survived more than 30 days with 1 or more posttransplant biopsy samples were included. Cox regression was used to examine the association between an HCV(+)D status and advanced fibrosis (stage 3/4 or higher). Ninety-nine of the 1206 patients (8%) received an HCV(+)D graft. Recipients of HCV(+)D grafts were older than recipients of hepatitis C virus antibody-negative donor [HCV(-)D] grafts (P = 0.03), but they were otherwise similar. HCV(+)D grafts were significantly lower in quality according to the donor risk index (P < 0.001). Advanced fibrosis occurred in 32% of HCV(+)D graft recipients and in 28% of HCV(-)D graft recipients (P = 0.39). The unadjusted 1- and 3-year rates of advanced fibrosis were significantly higher for HCV(+)D graft recipients (14% and 48%) versus HCV(-)D graft recipients (7% and 33%, P = 0.01). Transplantation with HCV(+)D grafts was associated with a 58% increased risk of advanced fibrosis [95% confidence interval (CI) = 1.05-2.36, P = 0.03]. However, in an analysis stratified by the mean donor age of 45 years, an HCV(+)D status was associated with advanced fibrosis only with donors >45 years old [hazard ratio (HR) = 1.76, 95% CI = 1.06-2.93, P = 0.03] and not with donors ≤45 years old (HR = 0.94, 95% CI = 0.47-1.87, P = 0.85). In conclusion, a careful consideration of the risks and benefits is needed with HCV(+)D grafts. Recipients of HCV(+)D grafts (especially from older donors) should undergo close monitoring for more rapidly progressive fibrosis. Studies are needed to determine whether early HCV therapy modifies this risk.
Copyright © 2012 American Association for the Study of Liver Diseases.
Figures



References
-
-
Based on Organ Procurement and Transplantation Network (OPTN) data as of February 26, 2010.
-
-
- Northup PG, Argo CK, Nguyen DT, McBride MA, Kumer SC, Schmitt TM, et al. Liver allografts from hepatitis C positive donors can offer good outcomes in hepatitis C positive recipients: a US National Transplant Registry analysis. Transpl Int. 2010 Apr 21; - PubMed
-
- Velidedeoglu E, Desai NM, Campos L, Olthoff KM, Shaked A, Nunes F, et al. The outcome of liver grafts procured from hepatitis C-positive donors. Transplantation. 2002 Feb 27;73(4):582–7. - PubMed
-
- Burr AT, Li Y, Tseng JF, Saidi RF, Bozorgzadeh A, Shah SA. Survival After Liver Transplantation Using Hepatitis C Virus-Positive Donor Allografts: Case-Controlled Analysis of the UNOS Database. World J Surg. 2011 Mar 8; - PubMed
-
- Khapra AP, Agarwal K, Fiel MI, Kontorinis N, Hossain S, Emre S, et al. Impact of donor age on survival and fibrosis progression in patients with hepatitis C undergoing liver transplantation using HCV+ allografts. Liver Transpl. 2006 Oct;12(10):1496–503. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
