Folding of the hammerhead ribozyme: pyrrolo-cytosine fluorescence separates core folding from global folding and reveals a pH-dependent conformational change
- PMID: 22274955
- PMCID: PMC3285932
- DOI: 10.1261/rna.030999.111
Folding of the hammerhead ribozyme: pyrrolo-cytosine fluorescence separates core folding from global folding and reveals a pH-dependent conformational change
Abstract
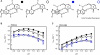
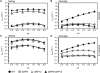
The catalytic activity of the hammerhead ribozyme is limited by its ability to fold into the native tertiary structure. Analysis of folding has been hampered by a lack of assays that can independently monitor the environment of nucleobases throughout the ribozyme-substrate complex in real time. Here, we report the development and application of a new folding assay in which we use pyrrolo-cytosine (pyC) fluorescence to (1) probe active-site formation, (2) examine the ability of peripheral ribozyme domains to support native folding, (3) identify a pH-dependent conformational change within the ribozyme, and (4) explore its influence on the equilibrium between the folded and unfolded core of the hammerhead ribozyme. We conclude that the natural ribozyme folds in two distinct noncooperative steps and the pH-dependent correlation between core folding and activity is linked to formation of the G8-C3 base pair.
Figures




References
-
- Berry DA, Jung K-Y, Wise DS, Sercel AD, Pearson WH, Mackie H, Randolph JB, Somers RL 2004. Pyrrolo-dC and pyrrolo-C: Fluorescent analogs of cytidine and 2′-deoxycytidine for the study of oligonucleotides. Tetrahedron Lett 45: 2457–2461
-
- Bevilacqua PC, Brown TS, Chadalavada D, Lecomte J, Moody E, Nakano SI 2005. Linkage between proton binding and folding in RNA: Implications for RNA catalysis. Biochem Soc Trans 33: 466–470 - PubMed
-
- Brahms J, Maurizot JC, Michelson AM 1967. Conformational stability of dinucleotides in solution. J Mol Biol 25: 481–495 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
