Stable incorporation of ATPase subunits into 19 S regulatory particle of human proteasome requires nucleotide binding and C-terminal tails
- PMID: 22275368
- PMCID: PMC3308751
- DOI: 10.1074/jbc.M111.316208
Stable incorporation of ATPase subunits into 19 S regulatory particle of human proteasome requires nucleotide binding and C-terminal tails
Abstract
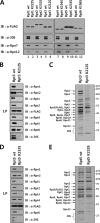
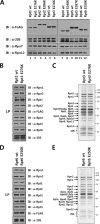
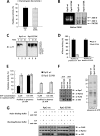
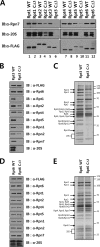
The 26 S proteasome is a large multi-subunit protein complex that degrades ubiquitinated proteins in eukaryotic cells. Proteasome assembly is a complex process that involves formation of six- and seven-membered ring structures from homologous subunits. Here we report that the assembly of hexameric Rpt ring of the 19 S regulatory particle (RP) requires nucleotide binding but not ATP hydrolysis. Disruption of nucleotide binding to an Rpt subunit by mutation in the Walker A motif inhibits the assembly of the Rpt ring without affecting heterodimer formation with its partner Rpt subunit. Coexpression of the base assembly chaperones S5b and PAAF1 with mutant Rpt1 and Rpt6, respectively, relieves assembly inhibition of mutant Rpts by facilitating their interaction with adjacent Rpt dimers. The mutation in the Walker B motif which impairs ATP hydrolysis does not affect Rpt ring formation. Incorporation of a Walker B mutant Rpt subunit abrogates the ATPase activity of the 19 S RP, suggesting that failure of the mutant Rpt to undergo the conformational transition from an ATP-bound to an ADP-bound state impairs conformational changes in the other five wild-type Rpts in the Rpt ring. In addition, we demonstrate that the C-terminal tails of Rpt subunits possessing core particle (CP)-binding affinities facilitate the cellular assembly of the 19 S RP, implying that the 20 S CP may function as a template for base assembly in human cells. Taken together, these results suggest that the ATP-bound conformational state of an Rpt subunit with the exposed C-terminal tail is competent for cellular proteasome assembly.
Figures



Similar articles
-
Assembly manual for the proteasome regulatory particle: the first draft.Biochem Soc Trans. 2010 Feb;38(Pt 1):6-13. doi: 10.1042/BST0380006. Biochem Soc Trans. 2010. PMID: 20074027 Free PMC article. Review.
-
Proteasome Activation is Mediated via a Functional Switch of the Rpt6 C-terminal Tail Following Chaperone-dependent Assembly.Sci Rep. 2015 Oct 9;5:14909. doi: 10.1038/srep14909. Sci Rep. 2015. PMID: 26449534 Free PMC article.
-
Hexameric assembly of the proteasomal ATPases is templated through their C termini.Nature. 2009 Jun 11;459(7248):866-70. doi: 10.1038/nature08065. Nature. 2009. PMID: 19412160 Free PMC article.
-
Reconfiguration of the proteasome during chaperone-mediated assembly.Nature. 2013 May 23;497(7450):512-6. doi: 10.1038/nature12123. Epub 2013 May 5. Nature. 2013. PMID: 23644457 Free PMC article.
-
Order of the proteasomal ATPases and eukaryotic proteasome assembly.Cell Biochem Biophys. 2011 Jun;60(1-2):13-20. doi: 10.1007/s12013-011-9178-4. Cell Biochem Biophys. 2011. PMID: 21461838 Free PMC article. Review.
Cited by
-
Alpha-ring Independent Assembly of the 20S Proteasome.Sci Rep. 2015 Aug 19;5:13130. doi: 10.1038/srep13130. Sci Rep. 2015. PMID: 26286114 Free PMC article.
-
Functional asymmetries of proteasome translocase pore.J Biol Chem. 2012 May 25;287(22):18535-43. doi: 10.1074/jbc.M112.357327. Epub 2012 Apr 5. J Biol Chem. 2012. PMID: 22493437 Free PMC article.
-
Structural basis for dynamic regulation of the human 26S proteasome.Proc Natl Acad Sci U S A. 2016 Nov 15;113(46):12991-12996. doi: 10.1073/pnas.1614614113. Epub 2016 Oct 21. Proc Natl Acad Sci U S A. 2016. PMID: 27791164 Free PMC article.
-
The proteasome-associated protein Ecm29 inhibits proteasomal ATPase activity and in vivo protein degradation by the proteasome.J Biol Chem. 2013 Oct 11;288(41):29467-81. doi: 10.1074/jbc.M113.491662. Epub 2013 Aug 30. J Biol Chem. 2013. PMID: 23995839 Free PMC article.
-
Nucleotide-dependent switch in proteasome assembly mediated by the Nas6 chaperone.Proc Natl Acad Sci U S A. 2017 Feb 14;114(7):1548-1553. doi: 10.1073/pnas.1612922114. Epub 2017 Jan 30. Proc Natl Acad Sci U S A. 2017. PMID: 28137839 Free PMC article.
References
-
- Schwartz A. L., Ciechanover A. (2009) Targeting proteins for destruction by the ubiquitin system: implications for human pathobiology. Annu. Rev. Pharmacol. Toxicol. 49, 73–96 - PubMed
-
- Groll M., Ditzel L., Löwe J., Stock D., Bochtler M., Bartunik H. D., Huber R. (1997) Structure of 20 S proteasome from yeast at 2.4 A resolution. Nature. 386, 463–471 - PubMed
-
- Whitby F. G., Masters E. I., Kramer L., Knowlton J. R., Yao Y., Wang C. C., Hill C. P. (2000) Structural basis for the activation of 20 S proteasomes by 11S regulators. Nature. 408, 115–120 - PubMed
-
- Groll M., Bajorek M., Köhler A., Moroder L., Rubin D. M., Huber R., Glickman M. H., Finley D. (2000) A gated channel into the proteasome core particle. Nat. Struct. Biol. 7, 1062–1067 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

