S100B engages RAGE or bFGF/FGFR1 in myoblasts depending on its own concentration and myoblast density. Implications for muscle regeneration
- PMID: 22276098
- PMCID: PMC3262793
- DOI: 10.1371/journal.pone.0028700
S100B engages RAGE or bFGF/FGFR1 in myoblasts depending on its own concentration and myoblast density. Implications for muscle regeneration
Abstract
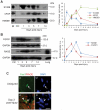
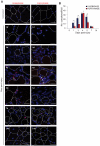
In high-density myoblast cultures S100B enhances basic fibroblast growth factor (bFGF) receptor 1 (FGFR1) signaling via binding to bFGF and blocks its canonical receptor, receptor for advanced glycation end-products (RAGE), thereby stimulating proliferation and inhibiting differentiation. Here we show that upon skeletal muscle injury S100B is released from myofibers with maximum release at day 1 post-injury in coincidence with satellite cell activation and the beginning of the myoblast proliferation phase, and declining release thereafter in coincidence with reduced myoblast proliferation and enhanced differentiation. By contrast, levels of released bFGF are remarkably low at day 1 post-injury, peak around day 5 and decline thereafter. We also show that in low-density myoblast cultures S100B binds RAGE, but not bFGF/FGFR1 thereby simultaneously stimulating proliferation via ERK1/2 and activating the myogenic program via p38 MAPK. Clearance of S100B after a 24-h treatment of low-density myoblasts results in enhanced myotube formation compared with controls as a result of increased cell numbers and activated myogenic program, whereas chronic treatment with S100B results in stimulation of proliferation and inhibition of differentiation due to a switch of the initial low-density culture to a high-density culture. However, at relatively high doses, S100B stimulates the mitogenic bFGF/FGFR1 signaling in low-density myoblasts, provided bFGF is present. We propose that S100B is a danger signal released from injured muscles that participates in skeletal muscle regeneration by activating the promyogenic RAGE or the mitogenic bFGF/FGFR1 depending on its own concentration, the absence or presence of bFGF, and myoblast density.
Conflict of interest statement
Figures





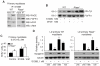

Similar articles
-
S100B protein regulates myoblast proliferation and differentiation by activating FGFR1 in a bFGF-dependent manner.J Cell Sci. 2011 Jul 15;124(Pt 14):2389-400. doi: 10.1242/jcs.084491. Epub 2011 Jun 21. J Cell Sci. 2011. PMID: 21693575
-
Amphoterin stimulates myogenesis and counteracts the antimyogenic factors basic fibroblast growth factor and S100B via RAGE binding.Mol Cell Biol. 2004 Jun;24(11):4880-94. doi: 10.1128/MCB.24.11.4880-4894.2004. Mol Cell Biol. 2004. PMID: 15143181 Free PMC article.
-
The amphoterin (HMGB1)/receptor for advanced glycation end products (RAGE) pair modulates myoblast proliferation, apoptosis, adhesiveness, migration, and invasiveness. Functional inactivation of RAGE in L6 myoblasts results in tumor formation in vivo.J Biol Chem. 2006 Mar 24;281(12):8242-53. doi: 10.1074/jbc.M509436200. Epub 2006 Jan 9. J Biol Chem. 2006. PMID: 16407300
-
S100B-stimulated NO production by BV-2 microglia is independent of RAGE transducing activity but dependent on RAGE extracellular domain.Biochim Biophys Acta. 2004 Dec 6;1742(1-3):169-77. doi: 10.1016/j.bbamcr.2004.09.008. Biochim Biophys Acta. 2004. PMID: 15590067 Review.
-
Involvement of phosphatidylserine receptors in the skeletal muscle regeneration: therapeutic implications.J Cachexia Sarcopenia Muscle. 2022 Aug;13(4):1961-1973. doi: 10.1002/jcsm.13024. Epub 2022 Jun 5. J Cachexia Sarcopenia Muscle. 2022. PMID: 35666022 Free PMC article. Review.
Cited by
-
Cellular and molecular mechanisms of sarcopenia: the S100B perspective.J Cachexia Sarcopenia Muscle. 2018 Dec;9(7):1255-1268. doi: 10.1002/jcsm.12363. Epub 2018 Nov 30. J Cachexia Sarcopenia Muscle. 2018. PMID: 30499235 Free PMC article. Review.
-
RAGE in the pathophysiology of skeletal muscle.J Cachexia Sarcopenia Muscle. 2018 Dec;9(7):1213-1234. doi: 10.1002/jcsm.12350. Epub 2018 Oct 18. J Cachexia Sarcopenia Muscle. 2018. PMID: 30334619 Free PMC article. Review.
-
How S100B crosses brain barriers and why it is considered a peripheral marker of brain injury.Exp Biol Med (Maywood). 2023 Nov;248(22):2109-2119. doi: 10.1177/15353702231214260. Epub 2023 Dec 6. Exp Biol Med (Maywood). 2023. PMID: 38058025 Free PMC article. Review.
-
S100B protein in tissue development, repair and regeneration.World J Biol Chem. 2013 Feb 26;4(1):1-12. doi: 10.4331/wjbc.v4.i1.1. World J Biol Chem. 2013. PMID: 23580916 Free PMC article.
-
RAGE and tobacco smoke: insights into modeling chronic obstructive pulmonary disease.Front Physiol. 2012 Jul 25;3:301. doi: 10.3389/fphys.2012.00301. eCollection 2012. Front Physiol. 2012. PMID: 22934052 Free PMC article.
References
-
- Chargé SB, Rudnicki MA. Cellular and molecular regulation of muscle regeneration. Physiol Rev. 2004;84:209–238. - PubMed
-
- Buckingham M. Myogenic progenitor cells and skeletal myogenesis in vertebrates. Curr Opin Genet Dev. 2006;16:525–532. - PubMed
-
- Donato R, Sorci G, Riuzzi F, Arcuri C, Bianchi R, et al. S100B's double life: intracellular regulator and extracellular signal. Biochm Biophys Acta. 2009;1793:1008–1022. - PubMed
-
- Bierhaus A, Humpert PM, Morcos M, Wendt T, Chavakis T, et al. Understanding RAGE, the receptor for advanced glycation end products. J Mol Med. 2005;83:876–886. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

