Probing amphotericin B single channel activity by membrane dipole modifiers
- PMID: 22276169
- PMCID: PMC3261894
- DOI: 10.1371/journal.pone.0030261
Probing amphotericin B single channel activity by membrane dipole modifiers
Abstract
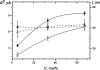
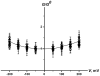
The effects of dipole modifiers and their structural analogs on the single channel activity of amphotericin B in sterol-containing planar phosphocholine membranes are studied. It is shown that the addition of phloretin in solutions bathing membranes containing cholesterol or ergosterol decreases the conductance of single amphotericin B channels. Quercetin decreases the channel conductance in cholesterol-containing bilayers while it does not affect the channel conductance in ergosterol-containing membranes. It is demonstrated that the insertion of styryl dyes, such as RH 421, RH 237 or RH 160, in bilayers with either cholesterol or ergosterol leads to the increase of the current amplitude of amphotericin B pores. Introduction of 5α-androstan-3β-ol into a membrane-forming solution increases the amphotericin B channel conductance in a concentration-dependent manner. All the effects are likely to be attributed to the influence of the membrane dipole potential on the conductance of single amphotericin B channels. However, specific interactions of some dipole modifiers with polyene-sterol complexes might also contribute to the activity of single amphotericin B pores. It has been shown that the channel dwell time increases with increasing sterol concentration, and it is higher for cholesterol-containing membranes than for bilayers including ergosterol, 6-ketocholestanol, 7-ketocholestanol or 5α-androstan-3β-ol. These findings suggest that the processes of association/dissociation of channel forming molecules depend on the membrane fluidity.
Conflict of interest statement
Figures
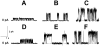

Similar articles
-
The interaction of dipole modifiers with polyene-sterol complexes.PLoS One. 2012;7(9):e45135. doi: 10.1371/journal.pone.0045135. Epub 2012 Sep 21. PLoS One. 2012. PMID: 23028805 Free PMC article.
-
The interaction of dipole modifiers with amphotericin-ergosterol complexes. Effects of phospholipid and sphingolipid membrane composition.Eur Biophys J. 2014 May;43(4-5):207-15. doi: 10.1007/s00249-014-0946-0. Epub 2014 Feb 23. Eur Biophys J. 2014. PMID: 24563224
-
Membrane dipole modifiers modulate single-length nystatin channels via reducing elastic stress in the vicinity of the lipid mouth of a pore.Biochim Biophys Acta. 2015 Jan;1848(1 Pt A):192-9. doi: 10.1016/j.bbamem.2014.09.004. Epub 2014 Sep 16. Biochim Biophys Acta. 2015. PMID: 25223717
-
Recent progress in the study of the interactions of amphotericin B with cholesterol and ergosterol in lipid environments.Eur Biophys J. 2014 Nov;43(10-11):453-67. doi: 10.1007/s00249-014-0983-8. Epub 2014 Aug 31. Eur Biophys J. 2014. PMID: 25173562 Free PMC article. Review.
-
[Antifungal macrocycle antibiotic amphotericin B - its present and future. Multidisciplinary perspective for the use in the medical practice].Biomed Khim. 2021 Jul;67(4):311-322. doi: 10.18097/PBMC20216704311. Biomed Khim. 2021. PMID: 34414889 Review. Russian.
Cited by
-
Cyclodextrins: Only Pharmaceutical Excipients or Full-Fledged Drug Candidates?Pharmaceutics. 2022 Nov 22;14(12):2559. doi: 10.3390/pharmaceutics14122559. Pharmaceutics. 2022. PMID: 36559052 Free PMC article. Review.
-
Polyene Antibiotics Physical Chemistry and Their Effect on Lipid Membranes; Impacting Biological Processes and Medical Applications.Membranes (Basel). 2022 Jun 30;12(7):681. doi: 10.3390/membranes12070681. Membranes (Basel). 2022. PMID: 35877884 Free PMC article. Review.
-
The influence of halogen derivatives of thyronine and fluorescein on the dipole potential of phospholipid membranes.J Membr Biol. 2014 Aug;247(8):739-45. doi: 10.1007/s00232-014-9703-7. Epub 2014 Jul 15. J Membr Biol. 2014. PMID: 25024118
-
Antibiotic Loaded Phytosomes as a Way to Develop Innovative Lipid Formulations of Polyene Macrolides.Pharmaceutics. 2024 May 16;16(5):665. doi: 10.3390/pharmaceutics16050665. Pharmaceutics. 2024. PMID: 38794328 Free PMC article.
-
The interaction of dipole modifiers with polyene-sterol complexes.PLoS One. 2012;7(9):e45135. doi: 10.1371/journal.pone.0045135. Epub 2012 Sep 21. PLoS One. 2012. PMID: 23028805 Free PMC article.
References
-
- Shigemi A, Matsumoto K, Ikawa K, Yaji K, Shimodozono Y, et al. Safety analysis of liposomal amphotericin B in adult patients: anaemia, thrombocytopenia, nephrotoxicity, hepatotoxicity and hypokalaemia. Int J Antimicrob Agent. 2011;30 In press. - PubMed
-
- Andreoli TE. The structure and function of amphotericin B-cholesterol pores in lipid bilayer membranes. Ann NY Acad Sci. 1974;235:448–468. - PubMed
-
- de Kruijff B, Gerritsen WJ, Oerlemans A, Demel RA, van Deenen LL. Polyene antibiotic-sterol interactions in membranes of Acholeplasma laidlawii cells and lecithin liposomes. I. Specificity of the membrane permeability changes induced by the polyene antibiotics. Biochim Biophys Acta. 1974;339:30–43. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

