Normalization of cholesterol homeostasis by 2-hydroxypropyl-β-cyclodextrin in neurons and glia from Niemann-Pick C1 (NPC1)-deficient mice
- PMID: 22277650
- PMCID: PMC3308731
- DOI: 10.1074/jbc.M111.326405
Normalization of cholesterol homeostasis by 2-hydroxypropyl-β-cyclodextrin in neurons and glia from Niemann-Pick C1 (NPC1)-deficient mice
Abstract
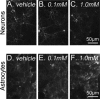
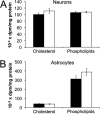
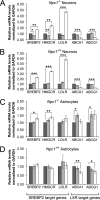
Niemann-Pick C (NPC) disease is an inherited, progressive neurodegenerative disorder caused by mutations in the NPC1 or NPC2 gene that result in an accumulation of unesterified cholesterol in late endosomes/lysosomes (LE/L) and impaired export of cholesterol from LE/L to the endoplasmic reticulum (ER). Recent studies demonstrate that administration of cyclodextrin (CD) to Npc1(-/-) mice eliminates cholesterol sequestration in LE/L of many tissues, including the brain, delays neurodegeneration, and increases lifespan of the mice. We have now investigated cholesterol homeostasis in NPC1-deficient cells of the brain in response to CD. Primary cultures of neurons and glial cells from Npc1(-/-) mice were incubated for 24 h with 0.1 to 10 mm CD after which survival and cholesterol homeostasis were monitored. Although 10 mm CD was profoundly neurotoxic, and altered astrocyte morphology, 0.1 and 1 mm CD were not toxic but effectively mobilized stored cholesterol from the LE/L as indicated by filipin staining. However, 0.1 and 1 mm CD altered cholesterol homeostasis in opposite directions. The data suggest that 0.1 mm CD releases cholesterol trapped in LE/L of neurons and astrocytes and increases cholesterol availability at the ER, whereas 1 mm CD primarily extracts cholesterol from the plasma membrane and reduces ER cholesterol. These studies in Npc1(-/-) neurons and astrocytes establish a dose of CD (0.1 mm) that would likely be beneficial in NPC disease. The findings are timely because treatment of NPC disease patients with CD is currently being initiated.
Figures








Similar articles
-
Cyclodextrin mediates rapid changes in lipid balance in Npc1-/- mice without carrying cholesterol through the bloodstream.J Lipid Res. 2012 Nov;53(11):2331-42. doi: 10.1194/jlr.M028241. Epub 2012 Aug 14. J Lipid Res. 2012. PMID: 22892156 Free PMC article.
-
Beneficial effects of primidone in Niemann-Pick disease type C (NPC)-model cells and mice: Reduction of unesterified cholesterol levels in cells and extension of lifespan in mice.Eur J Pharmacol. 2021 Apr 5;896:173907. doi: 10.1016/j.ejphar.2021.173907. Epub 2021 Jan 24. Eur J Pharmacol. 2021. PMID: 33503462
-
Hydroxypropyl-beta and -gamma cyclodextrins rescue cholesterol accumulation in Niemann-Pick C1 mutant cell via lysosome-associated membrane protein 1.Cell Death Dis. 2018 Oct 3;9(10):1019. doi: 10.1038/s41419-018-1056-1. Cell Death Dis. 2018. PMID: 30282967 Free PMC article.
-
Niemann-Pick C disease and mobilization of lysosomal cholesterol by cyclodextrin.J Lipid Res. 2014 Aug;55(8):1609-21. doi: 10.1194/jlr.R047837. Epub 2014 Mar 24. J Lipid Res. 2014. PMID: 24664998 Free PMC article. Review.
-
Function of the Niemann-Pick type C proteins and their bypass by cyclodextrin.Curr Opin Lipidol. 2011 Jun;22(3):204-9. doi: 10.1097/MOL.0b013e3283453e69. Curr Opin Lipidol. 2011. PMID: 21412152 Review.
Cited by
-
Reversal of Pathologic Lipid Accumulation in NPC1-Deficient Neurons by Drug-Promoted Release of LAMP1-Coated Lamellar Inclusions.J Neurosci. 2016 Jul 27;36(30):8012-25. doi: 10.1523/JNEUROSCI.0900-16.2016. J Neurosci. 2016. PMID: 27466344 Free PMC article.
-
Cyclodextrin mediates rapid changes in lipid balance in Npc1-/- mice without carrying cholesterol through the bloodstream.J Lipid Res. 2012 Nov;53(11):2331-42. doi: 10.1194/jlr.M028241. Epub 2012 Aug 14. J Lipid Res. 2012. PMID: 22892156 Free PMC article.
-
Hearing Loss and Otopathology Following Systemic and Intracerebroventricular Delivery of 2-Hydroxypropyl-Beta-Cyclodextrin.J Assoc Res Otolaryngol. 2015 Oct;16(5):599-611. doi: 10.1007/s10162-015-0528-6. Epub 2015 Jun 9. J Assoc Res Otolaryngol. 2015. PMID: 26055150 Free PMC article.
-
Single Cell Transcriptome Analysis of Niemann-Pick Disease, Type C1 Cerebella.Int J Mol Sci. 2020 Jul 28;21(15):5368. doi: 10.3390/ijms21155368. Int J Mol Sci. 2020. PMID: 32731618 Free PMC article.
-
Analytical Characterization of Methyl-β-Cyclodextrin for Pharmacological Activity to Reduce Lysosomal Cholesterol Accumulation in Niemann-Pick Disease Type C1 Cells.Assay Drug Dev Technol. 2017 May/Jun;15(4):154-166. doi: 10.1089/adt.2017.774. Assay Drug Dev Technol. 2017. PMID: 28631941 Free PMC article.
References
-
- Carstea E. D., Morris J. A., Coleman K. G., Loftus S. K., Zhang D., Cummings C., Gu J., Rosenfeld M. A., Pavan W. J., Krizman D. B., Nagle J., Polymeropoulos M. H., Sturley S. L., Ioannou Y. A., Higgins M. E., Comly M., Cooney A., Brown A., Kaneski C. R., Blanchette-Mackie E. J., Dwyer N. K., Neufeld E. B., Chang T. Y., Liscum L., Strauss J. F., 3rd, Ohno K., Zeigler M., Carmi R., Sokol J., Markie D., O'Neill R. R., van Diggelen O. P., Elleder M., Patterson M. C., Brady R. O., Vanier M. T., Pentchev P. G., Tagle D. A. (1997) Niemann-Pick C1 disease gene: Homology to mediators of cholesterol homeostasis. Science 277, 228–231 - PubMed
-
- Naureckiene S., Sleat D. E., Lackland H., Fensom A., Vanier M. T., Wattiaux R., Jadot M., Lobel P. (2000) Identification of HE1 as the second gene of Niemann-Pick C disease. Science 290, 2298–2301 - PubMed
-
- Vanier M. T., Millat G. (2003) Niemann-Pick disease type C. Clin. Genet. 64, 269–281 - PubMed
-
- Wojtanik K. M., Liscum L. (2003) The transport of low density lipoprotein-derived cholesterol to the plasma membrane is defective in NPC1 cells. J. Biol. Chem. 278, 14850–14856 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

