Glutathione reductase facilitates host defense by sustaining phagocytic oxidative burst and promoting the development of neutrophil extracellular traps
- PMID: 22279102
- PMCID: PMC3480216
- DOI: 10.4049/jimmunol.1102683
Glutathione reductase facilitates host defense by sustaining phagocytic oxidative burst and promoting the development of neutrophil extracellular traps
Abstract
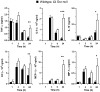
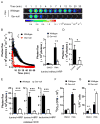
Glutathione reductase (Gsr) catalyzes the reduction of glutathione disulfide to glutathione, which plays an important role in the bactericidal function of phagocytes. Because Gsr has been implicated in the oxidative burst in human neutrophils and is abundantly expressed in the lymphoid system, we hypothesized that Gsr-deficient mice would exhibit marked defects during the immune response against bacterial challenge. We report in this study that Gsr-null mice exhibited enhanced susceptibility to Escherichia coli challenge, indicated by dramatically increased bacterial burden, cytokine storm, striking histological abnormalities, and substantially elevated mortality. Additionally, Gsr-null mice exhibited elevated sensitivity to Staphylococcus aureus. Examination of the bactericidal functions of the neutrophils from Gsr-deficient mice in vitro revealed impaired phagocytosis and defective bacterial killing activities. Although Gsr catalyzes the regeneration of glutathione, a major cellular antioxidant, Gsr-deficient neutrophils paradoxically produced far less reactive oxygen species upon activation both ex vivo and in vivo. Unlike wild-type neutrophils that exhibited a sustained oxidative burst upon stimulation with phorbol ester and fMLP, Gsr-deficient neutrophils displayed a very transient oxidative burst that abruptly ceased shortly after stimulation. Likewise, Gsr-deficient neutrophils also exhibited an attenuated oxidative burst upon encountering E. coli. Biochemical analysis revealed that the hexose monophosphate shunt was compromised in Gsr-deficient neutrophils. Moreover, Gsr-deficient neutrophils displayed a marked impairment in the formation of neutrophil extracellular traps, a bactericidal mechanism that operates after neutrophil death. Thus, Gsr-mediated redox regulation is crucial for bacterial clearance during host defense against massive bacterial challenge.
Figures

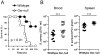





Similar articles
-
Glutathione reductase is essential for host defense against bacterial infection.Free Radic Biol Med. 2013 Aug;61:320-32. doi: 10.1016/j.freeradbiomed.2013.04.015. Epub 2013 Apr 24. Free Radic Biol Med. 2013. PMID: 23623936 Free PMC article.
-
Glutathione Reductase Promotes Fungal Clearance and Suppresses Inflammation during Systemic Candida albicans Infection in Mice.J Immunol. 2019 Oct 15;203(8):2239-2251. doi: 10.4049/jimmunol.1701686. Epub 2019 Sep 9. J Immunol. 2019. PMID: 31501257 Free PMC article.
-
Neutrophil-specific deletion of Syk kinase results in reduced host defense to bacterial infection.Blood. 2009 Nov 26;114(23):4871-82. doi: 10.1182/blood-2009-05-220806. Epub 2009 Oct 1. Blood. 2009. PMID: 19797524 Free PMC article.
-
Epic Immune Battles of History: Neutrophils vs. Staphylococcus aureus.Front Cell Infect Microbiol. 2017 Jun 30;7:286. doi: 10.3389/fcimb.2017.00286. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28713774 Free PMC article. Review.
-
Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance.Front Cell Infect Microbiol. 2017 Aug 25;7:373. doi: 10.3389/fcimb.2017.00373. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28890882 Free PMC article. Review.
Cited by
-
The emerging role for metabolism in fueling neutrophilic inflammation.Immunol Rev. 2023 Mar;314(1):427-441. doi: 10.1111/imr.13157. Epub 2022 Nov 3. Immunol Rev. 2023. PMID: 36326284 Free PMC article. Review.
-
Peptidylarginine Deiminase Inhibitor Suppresses Neutrophil Extracellular Trap Formation and MPO-ANCA Production.Front Immunol. 2016 Jun 8;7:227. doi: 10.3389/fimmu.2016.00227. eCollection 2016. Front Immunol. 2016. PMID: 27375623 Free PMC article.
-
Hyperoxidation of ether-linked phospholipids accelerates neutrophil extracellular trap formation.Sci Rep. 2017 Nov 22;7(1):16026. doi: 10.1038/s41598-017-15668-z. Sci Rep. 2017. PMID: 29167447 Free PMC article.
-
Regulation of Inflammatory Responses of Cow Mammary Epithelial Cells through MAPK Signaling Pathways of IL-17A Cytokines.Animals (Basel). 2024 May 25;14(11):1572. doi: 10.3390/ani14111572. Animals (Basel). 2024. PMID: 38891619 Free PMC article.
-
Molecular mechanisms regulating NETosis in infection and disease.Semin Immunopathol. 2013 Jul;35(4):513-30. doi: 10.1007/s00281-013-0384-6. Epub 2013 Jun 4. Semin Immunopathol. 2013. PMID: 23732507 Free PMC article. Review.
References
-
- Klebanoff SJ, Clark RA. The neutrophil: Function and clinical disorders. North-Holland Publishing Company; Amsterdam: 1978.
-
- Balija TM, Lowry SF. Lipopolysaccharide and sepsis-associated myocardial dysfunction. Curr Opin Infect Dis. 2011;24:248–253. - PubMed
-
- Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol. 2006;6:173–182. - PubMed
-
- Lambeth JD. NOX enzymes and the biology of reactive oxygen. Nat Rev Immunol. 2004;4:181–189. - PubMed
-
- Babior BM. NADPH oxidase. Curr Opin Immunol. 2004;16:42–47. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

