The effects of rosiglitazone on osteoblastic differentiation, osteoclast formation and bone resorption
- PMID: 22286232
- PMCID: PMC3887713
- DOI: 10.1007/s10059-012-2240-z
The effects of rosiglitazone on osteoblastic differentiation, osteoclast formation and bone resorption
Abstract
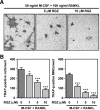
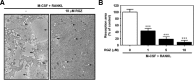
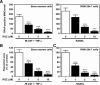
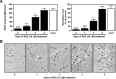
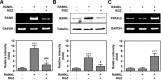
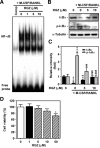
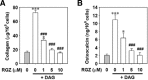
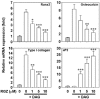
Rosiglitazone has the potential to activate peroxisome proliferator-activated receptor-γ (PPARγ), which in turn can affect bone formation and resorption. However, the mechanisms by which rosiglitazone regulates osteoclastic orosteoblastic differentiation are not fully understood. This study examines how rosiglitazone affects osteoclast formation, bone resorption and osteoblast differentiation from mouse bone marrow. Rosiglitazone treatment not only inhibited the formation of tartrate-resistant acid phosphatase-positive cells, but also prevented pit formation by bone marrow cells in a dose- and time-dependent manner. Rosiglitazone also suppressed the receptor activator of nuclear factor (NF)-κB ligand (RANKL) receptor(RANK) expression but increased PPARγ2 expression in the cells. In addition, rosiglitazone diminished RANKL induced activation of NF-κB-DNA binding by blocking IκBαphosphorylation. Furthermore, it reduced collagen and osteocalcin levels to nearly zero and prevented mRNA expression of osteoblast-specific proteins including runtrelated transcription factor-2, osteocalcin, and type I collagen.However, mRNA levels of adipocyte-specific marker, aP2, were markedly increased in the cells co-incubated with rosiglitazone. These results suggest that PPARγ activation by rosiglitazone inhibits osteoblast differentiation with increased adipogenesis in bone marrow cells and also may prevent osteoclast formation and bone resorptionin the cells.
Figures
References
-
- Ali A.A., Weinstein R.S., Stewart S.A., Parfitt A.M., Manolagas S.C., Jilka R.L. Rosiglitazone causes bone loss in mice by suppressing osteoblast differentiation and bone formation. Endocrinology. 2005;146:1226–1235. - PubMed
-
- Baek W.Y., Kim J.E. Transcriptional regulation of bone formation. Front. Biosci. (Schol Ed.) 2011;3:126–135. - PubMed
-
- Benvenuti S., Cellai I., Luciani P., Deledda C., Baglioni S., Giuliani C., Saccardi R., Mazzanti B., Dal Pozzo. S., Mannucci E., et al. Rosiglitazone stimulates adipogenesis and decreases osteoblastogenesis in human mesenchymal stem cells. J. Endocrinol. Invest. 2007;30:RC26–30. - PubMed
-
- Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

