Glioblastoma cancer stem-like cells: implications for pathogenesis and treatment
- PMID: 22290263
- PMCID: PMC3269658
- DOI: 10.1097/PPO.0b013e3182452e0d
Glioblastoma cancer stem-like cells: implications for pathogenesis and treatment
Abstract
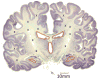
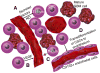
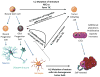
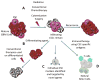
Glioblastoma remains one of the deadliest forms of cancer. Infiltrating cancer cells in the surrounding brain prevent complete resection, and tumor cell resistance to chemoradiation results in the poor prognosis of the glioblastoma (GBM) patient. Much research has been devoted over the years to the pathogenesis and treatment of GBM. The tumor stem cell hypothesis, which was initially described in hematopoietic cell malignancies, may explain the resistance of these tumors to conventional therapies. In this model, a certain subset of tumor cells, with characteristics similar to normal stem cells, is capable of producing the variety of cell types, which constitute the bulk of a tumor. As these tumor cells have properties distinct from those constituting the bulk of the tumor, a different approach may be required to eradicate these residual cells within the brain. Here we outline the history behind the theory of GBM cancer stem-like cells, as they are now referred to. We will also discuss the implications of their existence on commonly held beliefs about GBM pathogenesis and how they might influence future treatment strategies.
Figures
Similar articles
-
Heterogeneity of cancer-initiating cells within glioblastoma.Front Biosci (Schol Ed). 2012 Jun 1;4(4):1235-48. doi: 10.2741/s328. Front Biosci (Schol Ed). 2012. PMID: 22652868 Review.
-
Cancer Stem Cells: Significance in Origin, Pathogenesis and Treatment of Glioblastoma.Cells. 2021 Mar 11;10(3):621. doi: 10.3390/cells10030621. Cells. 2021. PMID: 33799798 Free PMC article. Review.
-
The three-layer concentric model of glioblastoma: cancer stem cells, microenvironmental regulation, and therapeutic implications.ScientificWorldJournal. 2011;11:1829-41. doi: 10.1100/2011/736480. Epub 2011 Oct 23. ScientificWorldJournal. 2011. PMID: 22125441 Free PMC article. Review.
-
Gliomagenesis: a game played by few players or a team effort?Front Biosci (Elite Ed). 2012 Jan 1;4(1):205-13. doi: 10.2741/e370. Front Biosci (Elite Ed). 2012. PMID: 22201865 Review.
-
Self‑renewal signaling pathways and differentiation therapies of glioblastoma stem cells (Review).Int J Oncol. 2021 Jul;59(1):45. doi: 10.3892/ijo.2021.5225. Epub 2021 May 20. Int J Oncol. 2021. PMID: 34013362
Cited by
-
The Transcription Regulator Krüppel-Like Factor 4 and Its Dual Roles of Oncogene in Glioblastoma and Tumor Suppressor in Neuroblastoma.For Immunopathol Dis Therap. 2016;7(1-2):127-139. doi: 10.1615/ForumImmunDisTher.2016017227. For Immunopathol Dis Therap. 2016. PMID: 28497005 Free PMC article.
-
Impact of Regorafenib on Endothelial Transdifferentiation of Glioblastoma Stem-like Cells.Cancers (Basel). 2022 Mar 18;14(6):1551. doi: 10.3390/cancers14061551. Cancers (Basel). 2022. PMID: 35326702 Free PMC article.
-
Inhibition of EGFR induces a c-MET-driven stem cell population in glioblastoma.Stem Cells. 2014 Feb;32(2):338-48. doi: 10.1002/stem.1554. Stem Cells. 2014. PMID: 24115218 Free PMC article.
-
Oncolytic herpes simplex virus-based strategies: toward a breakthrough in glioblastoma therapy.Front Microbiol. 2014 Jun 20;5:303. doi: 10.3389/fmicb.2014.00303. eCollection 2014. Front Microbiol. 2014. PMID: 24999342 Free PMC article. Review.
-
Immunosuppressive mechanisms in glioblastoma.Neuro Oncol. 2015 Nov;17 Suppl 7(Suppl 7):vii9-vii14. doi: 10.1093/neuonc/nov151. Neuro Oncol. 2015. PMID: 26516226 Free PMC article. Review.
References
-
- Brat DJ, Prayson RA, Ryken TC, et al. Diagnosis of malignant glioma: role of neuropathology. J Neurooncol. 2008;89(3):287–311. - PubMed
-
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–96. - PubMed
-
- Legler JM, Ries LA, Smith MA, et al. Cancer surveillance series [corrected]: brain and other central nervous system cancers: recent trends in incidence and mortality. J Natl Cancer Inst. 1999;91(16):1382–90. - PubMed
-
- Brem H, Piantadosi S, Burger PC, et al. Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet. 1995;345(8956):1008–12. - PubMed
-
- Vredenburgh JJ, Desjardins A, Herndon JE, 2nd, et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol. 2007;25(30):4722–9. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

