Three-dimensional and molecular analysis of the arterial pole of the developing human heart
- PMID: 22296102
- PMCID: PMC3375770
- DOI: 10.1111/j.1469-7580.2012.01474.x
Three-dimensional and molecular analysis of the arterial pole of the developing human heart
Abstract
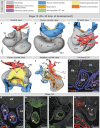
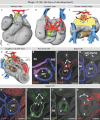
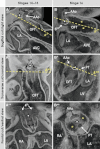
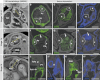
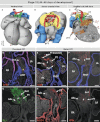
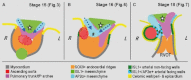
Labeling experiments in chicken and mouse embryos have revealed important roles for different cell lineages in the development of the cardiac arterial pole. These data can only fully be exploited when integrated into the continuously changing morphological context and compared with the patterns of gene expression. As yet, studies on the formation of separate ventricular outlets and arterial trunks in the human heart are exclusively based on histologically stained sections. So as to expand these studies, we performed immunohistochemical analyses of serially sectioned human embryos, along with three-dimensional reconstructions. The development of the cardiac arterial pole involves several parallel and independent processes of formation and fusion of outflow tract cushions, remodeling of the aortic sac and closure of an initial aortopulmonary foramen through formation of a transient aortopulmonary septum. Expression patterns of the transcription factors ISL1, SOX9 and AP2α show that, in addition to fusion of the SOX9-positive endocardial cushions, intrapericardial protrusion of pharyngeal mesenchyme derived from the neural crest contributes to the separation of the developing ascending aorta from the pulmonary trunk. The non-adjacent walls of the intrapericardial arterial trunks are formed through addition of ISL1-positive cells to the distal outflow tract, while the facing parts of the walls form from the protruding mesenchyme. The morphogenetic steps, along with the gene expression patterns reported in this study, are comparable to those observed in the mouse. They confirm the involvement of mesenchymal tissues derived from endocardium, mesoderm and migrating neural crest cells in the process of initial septation of the distal part of the outflow tract, and its subsequent separation into discrete intrapericardial arterial trunks.
© 2012 The Authors. Journal of Anatomy © 2012 Anatomical Society.
Figures


References
-
- Beall AC, Rosenquist TH. Smooth muscle cells of neural crest origin form the aorticopulmonary septum in the avian embryo. Anat Rec. 1990;226:360–366. - PubMed
-
- Bergwerff M, Verberne ME, DeRuiter MC, et al. Neural crest cell contribution to the developing circulatory system: implications for vascular morphology. Circ Res. 1998;82:221–231. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

