Carboxy-terminus of CXCR7 regulates receptor localization and function
- PMID: 22300987
- PMCID: PMC3294180
- DOI: 10.1016/j.biocel.2012.01.007
Carboxy-terminus of CXCR7 regulates receptor localization and function
Abstract
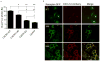
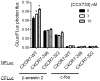
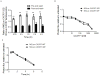
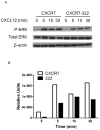
Chemokine receptor CXCR7 is essential for normal development, and this receptor promotes initiation and progression of diseases including cancer and autoimmunity. To understand normal and pathologic functions of CXCR7 and advance development of therapeutic agents, there is a need to define structural domains that regulate this receptor. We generated mutants of CXCR7 with deletion of different lengths of the predicted intracellular tail and analyzed effects on CXCR7 signaling and function in cell-based assays. While wild-type CXCR7 predominantly localized to intracellular vesicles, progressive deletion of the carboxy terminus redistributed the receptor to the plasma membrane. Truncating the intracellular tail of CXCR7 did not alter binding to CXCL12, but mutant receptors had reduced scavenging of this chemokine. Using a firefly luciferase complementation system, we established that deletions of the carboxy terminus decreased basal interactions and eliminated ligand-dependent recruitment of the scaffolding protein β-arrestin-2 to receptors. Deleting the carboxy terminus of CXCR7 impaired constitutive internalization of the receptor and reduced activation of ERK1/2 by CXCL12-CXCR7. Inhibiting dynamin, a molecule required for internalization of CXCR7, increased ligand-dependent association of the receptor with β-arrestin-2 and enhanced activation of ERK1/2. These studies establish mechanisms of action for CXCR7 and establish the intracellular tail of CXCR7 as a critical determinant of receptor trafficking, chemokine scavenging, and signaling.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures




References
-
- Boldajipour B, Mahabaleshwar S, Kardash E, Reichman-Fried M, Blaser H, Minina S, Wilson D, Xu Q, Raz E. Control of chemokine-guided cell migration by ligand sequestration. Cell. 2008;132:463–473. - PubMed
-
- Cen B, Yu Q, Guo J, Wu Y, Ling K, Cheng Z, Ma L, Pei G. Direct binding of beta-arrestins to two distinct intracellular domains of the delta opioid receptor. J Neurochem. 2001;76:1887–1894. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

