Development of an electrochemical immunoassay for detection of gatifloxacin in swine urine
- PMID: 22302425
- PMCID: PMC3274739
- DOI: 10.1631/jzus.B1100073
Development of an electrochemical immunoassay for detection of gatifloxacin in swine urine
Abstract
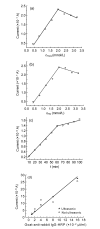
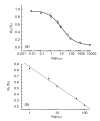
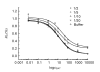
To detect gatifloxacin (GAT) residue in swine urine, an electrochemical immunoassay was established. An indirect competitive immunoassay was developed, in which the coating antigen is immobilized in an enzyme-linked immunosorbent assay (ELISA) plate and GAT residue from the sample competes with the limited binding sites in added anti-GAT antibody. Horseradish peroxidase (HRP) conjugated to goat anti-rabbit IgG was used as the enzymatic label. A carbon fiber working electrode was constructed and current signals were detected by using hydrogen peroxide as a substrate and hydroquinone as an electrochemical mediator. The electrochemical immunoassay was evaluated by analysis of GAT in buffer or swine urine and an average value of half inhibition concentration (IC(50)) of 8.9 ng/ml was obtained. Excellent specificity of the antibody was achieved with little cross-reaction with lomefloxacin (3.0%), ciprofloxacin (3.0%), and ofloxacin (1.9%) among commonly used (fluoro)quinolones. In conclusion, the immunoassay system developed in this research can be used as a rapid, powerful and on-site analytical tool to detect GAT residue in foods and food products.
Figures

Similar articles
-
Preparation of anti-gatifloxacin antibody and development of an indirect competitive enzyme-linked immunosorbent assay for the detection of gatifloxacin residue in milk.J Agric Food Chem. 2007 Aug 22;55(17):6879-84. doi: 10.1021/jf070978g. Epub 2007 Jul 24. J Agric Food Chem. 2007. PMID: 17645343
-
Electrochemical immunoassay using magnetic beads for the determination of zearalenone in baby food: an anticipated analytical tool for food safety.Anal Chim Acta. 2009 Oct 27;653(2):167-72. doi: 10.1016/j.aca.2009.09.024. Epub 2009 Sep 22. Anal Chim Acta. 2009. PMID: 19808109
-
Electrochemical adsorptive behavior of some fluoroquinolones at carbon paste electrode.Anal Sci. 2005 Oct;21(10):1249-54. doi: 10.2116/analsci.21.1249. Anal Sci. 2005. PMID: 16270589
-
Competitive electrochemical immunosensor for maduramicin detection by multiple signal amplification strategy via hemin@Fe-MIL-88NH2/AuPt.Biosens Bioelectron. 2019 Oct 1;142:111554. doi: 10.1016/j.bios.2019.111554. Epub 2019 Jul 31. Biosens Bioelectron. 2019. PMID: 31382098
-
Status of Physicochemical and Microbiological Analytical Methods of Gatifloxacin: A Review.J AOAC Int. 2022 Oct 26;105(6):1548-1554. doi: 10.1093/jaoacint/qsac089. J AOAC Int. 2022. PMID: 35861368 Review.
Cited by
-
Ultrasensitive Lateral Flow Immunoassay of Fluoroquinolone Antibiotic Gatifloxacin Using Au@Ag Nanoparticles as a Signal-Enhancing Label.Biosensors (Basel). 2024 Dec 6;14(12):598. doi: 10.3390/bios14120598. Biosensors (Basel). 2024. PMID: 39727863 Free PMC article.
-
A molecularly imprinted electrochemical sensor with dual functional monomers for selective determination of gatifloxacin.Mikrochim Acta. 2023 Jun 15;190(7):261. doi: 10.1007/s00604-023-05839-3. Mikrochim Acta. 2023. PMID: 37322368
References
-
- Blanco MH, Quintana LH, Hernandez L. Determination of dihydrozeatin riboside in apples by anodic stripping voltammetry with a carbon fiber microelectrode. Fresenius J Anal Chem. 1999;364(3):254–260. doi: 10.1007/s002160051333. - DOI
-
- Blondeau JM, Laskowski R, Bjarnason J, Stewart C. Comparative in vitro activity of gatifloxacin, grepafloxacin, levofloxacin, moxifloxacin and trovafloxacin against Gram-negative and Gram-positive organisms. Int J Antimicrob Agents. 2000;14(1):45–50. doi: 10.1016/S0924-8579(99)00143-0. - DOI - PubMed
-
- Conneely G, O′Mahony D, Lu H, Guilbault GG, Pravda M, Aherne M. An immunosensor for the detection of stanozolol in bovine urine. Anal Lett. 2007;40(7):1280–1293. doi: 10.1080/00032710701326650. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

