Arabidopsis thaliana: A Model for the Study of Root and Shoot Gravitropism
- PMID: 22303208
- PMCID: PMC3243349
- DOI: 10.1199/tab.0043
Arabidopsis thaliana: A Model for the Study of Root and Shoot Gravitropism
Abstract
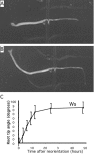
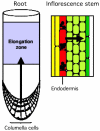
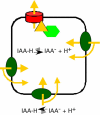
For most plants, shoots grow upward and roots grow downward. These growth patterns illustrate the ability for plant organs to guide their growth at a specified angle from the gravity vector (gravitropism). They allow shoots to grow upward toward light, where they can photosynthesize, and roots to grow downward into the soil, where they can anchor the plant as well as take up water and mineral ions.Gravitropism involves several steps organized in a specific response pathway. These include the perception of a gravistimulus (reorientation within the gravity field), the transduction of this mechanical stimulus into a physiological signal, the transmission of this signal from the site of sensing to the site of response, and a curvature-response which allows the organ tip to resume growth at a predefined set angle from the gravity vector.The primary sites for gravity sensing are located in the cap for roots, and in the endodermis for shoots. The curvature response occurs in the elongation zones for each organ. Upon gravistimulation, a gradient of auxin appears to be generated across the stimulated organ, and be transmitted to the site of response where it promotes a differential growth response. Therefore, while the gravity-induced auxin gradient has to be transmitted from the cap to the elongation zones in roots, there is no need for a longitudinal transport in shoots, as sites for gravity sensing and response overlap in this organ.A combination of molecular genetics, physiology, biochemistry and cell biology, coupled with the utilization of Arabidopsis thaliana as a model system, have recently allowed the identification of a number of molecules involved in the regulation of each phase of gravitropism in shoots and roots of higher plants. In this review, we attempt to summarize the results of these experiments, and we conclude by comparing the molecular and physiological mechanisms that underlie gravitropism in these organs.
Abbreviations: GSPA: gravitational set point angle; IAA: indole-3-acetic acid; NAA: 1-naphthalene acetic acid; NPA: 1-N-naphthylphthalamic acid; 2,4-D: 2,4-dichlorphenoxy acetic acid; TIBA: 2,3,5-triiodobenzoic acid.
Figures



Similar articles
-
Cellular and physiological functions of SGR family in gravitropic response in higher plants.J Adv Res. 2025 Jan;67:43-60. doi: 10.1016/j.jare.2024.01.026. Epub 2024 Feb 1. J Adv Res. 2025. PMID: 38295878 Free PMC article. Review.
-
Root gravitropism: an experimental tool to investigate basic cellular and molecular processes underlying mechanosensing and signal transmission in plants.Annu Rev Plant Biol. 2002;53:421-47. doi: 10.1146/annurev.arplant.53.100301.135158. Annu Rev Plant Biol. 2002. PMID: 12221983 Review.
-
Complex physiological and molecular processes underlying root gravitropism.Plant Mol Biol. 2002 Jun-Jul;49(3-4):305-17. Plant Mol Biol. 2002. PMID: 12036256 Review.
-
Molecular genetics of root gravitropism and waving in Arabidopsis thaliana.Gravit Space Biol Bull. 1998 May;11(2):71-8. Gravit Space Biol Bull. 1998. PMID: 11540641
-
Basipetal auxin transport is required for gravitropism in roots of Arabidopsis.Plant Physiol. 2000 Feb;122(2):481-90. doi: 10.1104/pp.122.2.481. Plant Physiol. 2000. PMID: 10677441 Free PMC article.
Cited by
-
Interaction with gravitropism, reversibility and lateral movements of phototropically stimulated potato shoots.J Plant Res. 2016 Jul;129(4):759-770. doi: 10.1007/s10265-016-0821-4. Epub 2016 Mar 31. J Plant Res. 2016. PMID: 27033355 Free PMC article.
-
Wortmannin-induced vacuole fusion enhances amyloplast dynamics in Arabidopsis zigzag1 hypocotyls.J Exp Bot. 2016 Dec;67(22):6459-6472. doi: 10.1093/jxb/erw418. Epub 2016 Nov 5. J Exp Bot. 2016. PMID: 27816929 Free PMC article.
-
The gravitropism defective 2 mutants of Arabidopsis are deficient in a protein implicated in endocytosis in Caenorhabditis elegans.Plant Physiol. 2004 Oct;136(2):3095-103; discussion 3002. doi: 10.1104/pp.104.050583. Epub 2004 Oct 1. Plant Physiol. 2004. PMID: 15466218 Free PMC article.
-
Cellular and physiological functions of SGR family in gravitropic response in higher plants.J Adv Res. 2025 Jan;67:43-60. doi: 10.1016/j.jare.2024.01.026. Epub 2024 Feb 1. J Adv Res. 2025. PMID: 38295878 Free PMC article. Review.
-
ALTERED RESPONSE TO GRAVITY is a peripheral membrane protein that modulates gravity-induced cytoplasmic alkalinization and lateral auxin transport in plant statocytes.Plant Cell. 2003 Nov;15(11):2612-25. doi: 10.1105/tpc.015560. Epub 2003 Sep 24. Plant Cell. 2003. PMID: 14507996 Free PMC article.
References
-
- Abel S., Nguyen M., Theologis A. The PS-IAA4/5-like family of early auxin-inducible mRNAs in Arabidopsis thaliana. J Mol Biol. 1995;2511(1):533–549. - PubMed
-
- Baluska F., Kreibaum A., Vitha S., Parker J., Barlow P., Sievers A. Central root cap cells are depleted of endoplasmic microtubules and actin microfilament bundles: Implications for their role as gravity sensing statocytes. Protoplasma. 1995;1961(1):212–223. - PubMed
-
- Bell C., Maher E. Mutants of Arabidopsis thaliana with abnormal gravitropic response. Mol Gen Genet. 1990;2201(1):289–293.
-
- Bennett M., Marchant A., Green H., May S., Ward S., Millner P., Walker A., Schulz B., Feldmann K. Arabidopsis AUX1 gene: a permease-like regulator of root gravitropism. Science. 1996;2731(1):948–950. - PubMed
-
- Bevan M., Mayer K., White O., Eisen J., Preuss D., Bureau T., Salzburg S., Mewes H. Sequence and Analysis of the Arabidopsis Genome. Curr Opin Plant Biol. 2001;41(1):105–110. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
