Nipah virus infects specific subsets of porcine peripheral blood mononuclear cells
- PMID: 22303463
- PMCID: PMC3267752
- DOI: 10.1371/journal.pone.0030855
Nipah virus infects specific subsets of porcine peripheral blood mononuclear cells
Abstract
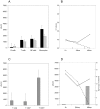
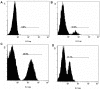
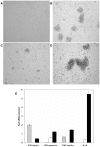
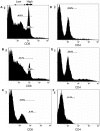
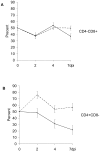
Nipah virus (NiV), a zoonotic paramyxovirus, is highly contagious in swine, and can cause fatal infections in humans following transmission from the swine host. The main viral targets in both species are the respiratory and central nervous systems, with viremia implicated as a mode of dissemination of NiV throughout the host. The presented work focused on the role of peripheral blood mononuclear cells (PBMC) in the viremic spread of the virus in the swine host. B lymphocytes, CD4-CD8-, as well as CD4+CD8- T lymphocytes were not permissive to NiV, and expansion of the CD4+CD8- cells early post infection was consistent with functional humoral response to NiV infection observed in swine. In contrast, significant drop in the CD4+CD8- T cell frequency was observed in piglets which succumbed to the experimental infection, supporting the hypothesis that antibody development is the critical component of the protective immune response. Productive viral replication was detected in monocytes, CD6+CD8+ T lymphocytes and NK cells by recovery of infectious virus in the cell supernatants. Virus replication was supported by detection of the structural N and the non-structural C proteins or by detection of genomic RNA increase in the infected cells. Infection of T cells carrying CD6 marker, a strong ligand for the activated leukocyte cell adhesion molecule ALCAM (CD166) highly expressed on the microvascular endothelial cell of the blood-air and the blood-brain barrier may explain NiV preferential tropism for small blood vessels of the lung and brain.
Conflict of interest statement
Figures


References
-
- Wang L, Harcourt BH, Yu M, Tamin A, Rota PA, et al. Molecular biology of Hendra and Nipah viruses. Microbes Infect. 2001;3:279–287. - PubMed
-
- Chua KB. Nipah virus outbreak in Malaysia. J Clin Virol. 2003;26:265–275. - PubMed
-
- Hossain MJ, Gurley ES, Montgomery JM, Bell M, Carroll DS, et al. Clinical presentation of Nipah virus infection in Bangladesh. Clin Infect Dis. 2008;46:977–984. - PubMed
-
- Chua KB, Bellini WJ, Rota PA, Harcourt BH, Tamin A, et al. Nipah virus: a recently emergent deadly paramyxovirus. Science. 2000;288:1432–1435. - PubMed
-
- Mohd Nor MN, Gan CH, Ong BL. Nipah virus infection of pigs in peninsular Malaysia. Rev Sci Tech. 2000;19:160–165. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

