Genistein as a potential inducer of the anti-atherogenic enzyme paraoxonase-1: studies in cultured hepatocytes in vitro and in rat liver in vivo
- PMID: 22304296
- PMCID: PMC3823426
- DOI: 10.1111/j.1582-4934.2012.01542.x
Genistein as a potential inducer of the anti-atherogenic enzyme paraoxonase-1: studies in cultured hepatocytes in vitro and in rat liver in vivo
Erratum in
- J Cell Mol Med. 2014 Aug;18(8):1704
Abstract
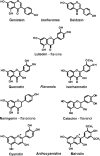
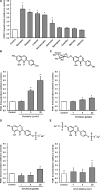
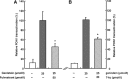
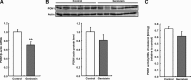
A number of cardioprotective effects, including the reduced oxidation of the low-density lipoprotein (LDL) particles, have been attributed to dietary soy isoflavones. Paraoxonase 1 (PON1), an enzyme mainly synthesized in the liver, may exhibit anti-atherogenic activity by protecting LDL from oxidation. Thus, dietary and pharmacological inducers of PON1 may decrease cardiovascular disease risk. Using a luciferase reporter gene assay we screened different flavonoids for their ability to induce PON1 in Huh7 hepatocytes in culture. Genistein was the most potent flavonoid with regard to its PON1-inducing activity, followed by daidzein, luteolin, isorhamnetin and quercetin. Other flavonoids such as naringenin, cyanidin, malvidin and catechin showed only little or no PON1-inducing activity. Genistein-mediated PON1 transactivation was partly inhibited by the oestrogen-receptor antagonist fulvestrant as well as by the aryl hydrocarbon receptor antagonist 7-ketocholesterol. In contrast to genistein, the conjugated genistein metabolites genistein-7-glucuronide, genistein-7-sulfate and genistein-7,4'-disulfate were only weak inducers of PON1 transactivation. Accordingly, dietary genistein supplementation (2 g/kg diet over three weeks) in growing rats did not increase hepatic PON1 mRNA and protein levels as well as plasma PON1 activity. Thus, genistein may be a PON1 inducer in cultured hepatocytes in vitro, but not in rats in vivo.
© 2012 The Authors Journal of Cellular and Molecular Medicine © 2012 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures


Similar articles
-
Curcumin induces paraoxonase 1 in cultured hepatocytes in vitro but not in mouse liver in vivo.Br J Nutr. 2011 Jan;105(2):167-70. doi: 10.1017/S0007114510004356. Epub 2010 Nov 16. Br J Nutr. 2011. PMID: 21078213
-
Quercetin up-regulates paraoxonase 1 gene expression with concomitant protection against LDL oxidation.Biochem Biophys Res Commun. 2009 Feb 20;379(4):1001-4. doi: 10.1016/j.bbrc.2009.01.015. Epub 2009 Jan 12. Biochem Biophys Res Commun. 2009. PMID: 19141295
-
Allyl isothiocyanate as a potential inducer of paraoxonase-1--studies in cultured hepatocytes and in mice.IUBMB Life. 2012 Feb;64(2):162-8. doi: 10.1002/iub.587. Epub 2011 Nov 30. IUBMB Life. 2012. PMID: 22131196
-
Paraoxonase, a cardioprotective enzyme: continuing issues.Curr Opin Lipidol. 2004 Jun;15(3):261-7. doi: 10.1097/00041433-200406000-00005. Curr Opin Lipidol. 2004. PMID: 15166781 Review.
-
Pharmacological and dietary modulators of paraoxonase 1 (PON1) activity and expression: the hunt goes on.Biochem Pharmacol. 2011 Feb 1;81(3):337-44. doi: 10.1016/j.bcp.2010.11.008. Epub 2010 Nov 18. Biochem Pharmacol. 2011. PMID: 21093416 Free PMC article. Review.
Cited by
-
The Search for Dietary Supplements to Elevate or Activate Circulating Paraoxonases.Int J Mol Sci. 2017 Feb 15;18(2):416. doi: 10.3390/ijms18020416. Int J Mol Sci. 2017. PMID: 28212288 Free PMC article.
-
Biochemical effects, hypolipidemic and anti-inflammatory activities of Artemisia vulgaris extract in hypercholesterolemic rats.J Clin Biochem Nutr. 2015 Jul;57(1):33-8. doi: 10.3164/jcbn.14-141. Epub 2015 Apr 18. J Clin Biochem Nutr. 2015. PMID: 26236098 Free PMC article.
-
Modulatory Effect of Lifestyle-Related, Environmental and Genetic Factors on Paraoxonase-1 Activity: A Review.Int J Environ Res Public Health. 2023 Feb 5;20(4):2813. doi: 10.3390/ijerph20042813. Int J Environ Res Public Health. 2023. PMID: 36833509 Free PMC article. Review.
-
Soy Extract, Rich in Hydroxylated Isoflavones, Exhibits Antidiabetic Properties In Vitro and in Drosophila melanogaster In Vivo.Nutrients. 2023 Mar 14;15(6):1392. doi: 10.3390/nu15061392. Nutrients. 2023. PMID: 36986122 Free PMC article.
-
Exploring the In Vivo Existence Forms (23 Original Constituents and 147 Metabolites) of Astragali Radix Total Flavonoids and Their Distributions in Rats Using HPLC-DAD-ESI-IT-TOF-MSn.Molecules. 2020 Nov 26;25(23):5560. doi: 10.3390/molecules25235560. Molecules. 2020. PMID: 33256251 Free PMC article.
References
-
- Poulter N. Coronary heart disease is a multifactorial disease. AJH. 1999;12:92S–5S. - PubMed
-
- Adlercreutz H, Mazur W. Phyto-oestrogens and Western diseases. Ann Med. 1997;29:95–120. - PubMed
-
- WHO. The global burden of disease: 2004 update. Geneva: World Health Organization; 2008.
-
- Beaglehole R. International trends in coronary heart disease mortality, morbidity, and risk factors. Epidemiol Rev. 1990;12:1–15. - PubMed
-
- Pilsakova L, Riecansky I, Jagla F. The physiological actions of isoflavone phytoestrogens. Physiol Res. 2010;59:651–64. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

