Characterization of Esterase A, a Pseudomonas stutzeri A15 Autotransporter
- PMID: 22307303
- PMCID: PMC3318783
- DOI: 10.1128/AEM.07690-11
Characterization of Esterase A, a Pseudomonas stutzeri A15 Autotransporter
Abstract
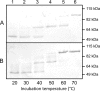
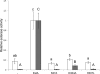
Autotransporters are a widespread family of proteins, generally known as virulence factors produced by Gram-negative bacteria. In this study, the esterase A (EstA) autotransporter of the rice root-colonizing beneficial bacterium Pseudomonas stutzeri A15 was characterized. A multiple sequence alignment identified EstA as belonging to clade II of the GDSL esterase family. Autologous overexpression allowed the investigation of several features of both autotransporter proteins and GDSL esterases. First, the correctly folded autotransporter was shown to be present in the membrane fraction. Unexpectedly, after separation of the membrane fraction, EstA was detected in the N-laurylsarcosine soluble fraction. However, evidence is presented for the surface exposure of EstA based on fluorescent labeling with EstA specific antibodies. Another remarkable feature is the occurrence of a C-terminal leucine residue instead of the canonical phenylalanine or tryptophan residue. Replacement of this residue with a phenylalanine residue reduced the stability of the β-barrel. Regarding the esterase passenger domain, we show the importance of the catalytic triad residues, with the serine and histidine residues being more critical than the aspartate residue. Furthermore, the growth of an estA-negative mutant was not impaired and cell mobility was not disabled compared to the wild type. No specific phenotype was detected for an estA-negative mutant. Overall, P. stutzeri A15 EstA is a new candidate for the surface display of proteins in environmentally relevant biotechnological applications.
Figures






Similar articles
-
Probing the applicability of autotransporter based surface display with the EstA autotransporter of Pseudomonas stutzeri A15.Microb Cell Fact. 2012 Dec 13;11:158. doi: 10.1186/1475-2859-11-158. Microb Cell Fact. 2012. PMID: 23237539 Free PMC article.
-
A novel lipolytic enzyme located in the outer membrane of Pseudomonas aeruginosa.J Bacteriol. 1999 Nov;181(22):6977-86. doi: 10.1128/JB.181.22.6977-6986.1999. J Bacteriol. 1999. PMID: 10559163 Free PMC article.
-
Use of Pseudomonas putida EstA as an anchoring motif for display of a periplasmic enzyme on the surface of Escherichia coli.Appl Environ Microbiol. 2004 Dec;70(12):6968-76. doi: 10.1128/AEM.70.12.6968-6976.2004. Appl Environ Microbiol. 2004. PMID: 15574889 Free PMC article.
-
Autotransporters with GDSL passenger domains: molecular physiology and biotechnological applications.Chembiochem. 2011 Jul 4;12(10):1476-85. doi: 10.1002/cbic.201100013. Epub 2011 May 19. Chembiochem. 2011. PMID: 21598370 Review.
-
Autotransporter secretion: varying on a theme.Res Microbiol. 2013 Jul-Aug;164(6):562-82. doi: 10.1016/j.resmic.2013.03.010. Epub 2013 Apr 6. Res Microbiol. 2013. PMID: 23567321 Review.
Cited by
-
Cell surface display of cold-active esterase EstPc with the use of a new autotransporter from Psychrobacter cryohalolentis K5(T).Extremophiles. 2015 Jan;19(1):161-70. doi: 10.1007/s00792-014-0695-0. Epub 2014 Sep 25. Extremophiles. 2015. PMID: 25253411
-
The intracellular citrus huanglongbing bacterium, 'Candidatus Liberibacter asiaticus' encodes two novel autotransporters.PLoS One. 2013 Jul 11;8(7):e68921. doi: 10.1371/journal.pone.0068921. Print 2013. PLoS One. 2013. PMID: 23874813 Free PMC article.
-
Microbial esterases and ester prodrugs: An unlikely marriage for combating antibiotic resistance.Drug Dev Res. 2019 Feb;80(1):33-47. doi: 10.1002/ddr.21468. Epub 2018 Oct 10. Drug Dev Res. 2019. PMID: 30302779 Free PMC article. Review.
-
Autotransporter domain-dependent enzymatic analysis of a novel extremely thermostable carboxylesterase with high biodegradability towards pyrethroid pesticides.Sci Rep. 2017 Jun 14;7(1):3461. doi: 10.1038/s41598-017-03561-8. Sci Rep. 2017. PMID: 28615636 Free PMC article.
-
Novel phenolic antimicrobials enhanced activity of iminodiacetate prodrugs against biofilm and planktonic bacteria.Chem Biol Drug Des. 2021 Jan;97(1):134-147. doi: 10.1111/cbdd.13768. Epub 2020 Sep 22. Chem Biol Drug Des. 2021. PMID: 32844569 Free PMC article.
References
-
- Akoh CC, Lee GC, Liaw YC, Huang TH, Shaw JF. 2004. GDSL family of serine esterases/lipases. Prog. Lipid Res. 43:534–552 - PubMed
-
- Asler IL, et al. 2010. Probing enzyme promiscuity of SGNH hydrolases. Chembiochem 11:2158–2167 - PubMed
-
- Becker S, et al. 2005. A generic system for the Escherichia coli cell-surface display of lipolytic enzymes. FEBS Lett. 579:1177–1182 - PubMed
-
- Bornscheuer UT. 2002. Microbial carboxyl esterases: classification, properties and application in biocatalysis. FEMS Microbiol. Rev. 26:73–81 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

