Vascular endothelial growth factor enhances macrophage clearance of apoptotic cells
- PMID: 22307908
- PMCID: PMC3330764
- DOI: 10.1152/ajplung.00116.2011
Vascular endothelial growth factor enhances macrophage clearance of apoptotic cells
Abstract
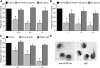
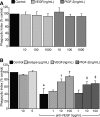
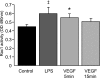
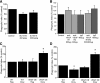
Efficient clearance of apoptotic cells from the lung by alveolar macrophages is important for the maintenance of tissue structure and function. Lung tissue from humans with emphysema contains increased numbers of apoptotic cells and decreased levels of vascular endothelial growth factor (VEGF). Mice treated with VEGF receptor inhibitors have increased numbers of apoptotic cells and develop emphysema. We hypothesized that VEGF regulates apoptotic cell clearance by alveolar macrophages (AM) via its interaction with VEGF receptor 1 (VEGF R1). Our data show that the uptake of apoptotic cells by murine AMs and human monocyte-derived macrophages is inhibited by depletion of VEGF and that VEGF activates Rac1. Antibody blockade or pharmacological inhibition of VEGF R1 activity also decreased apoptotic cell uptake ex vivo. Conversely, overexpression of VEGF significantly enhanced apoptotic cell uptake by AMs in vivo. These results indicate that VEGF serves a positive regulatory role via its interaction with VEGF R1 to activate Rac1 and enhance AM apoptotic cell clearance.
Figures
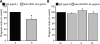
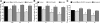
References
-
- Adini A, Kornaga T, Firoozbakht F, Benjamin LE. Placental growth factor is a survival factor for tumor endothelial cells and macrophages. Cancer Res 62: 2749– 2752, 2002 - PubMed
-
- Bhandari V, Choo-Wing R, Chapoval SP, Lee CG, Tang C, Kim YK, Ma B, Baluk P, Lin MI, McDonald DM, Homer RJ, Sessa WC, Elias JA. Essential role of nitric oxide in VEGF-induced, asthma-like angiogenic, inflammatory, mucus, and physiologic responses in the lung. Proc Natl Acad Sci USA 103: 11021– 11026, 2006 - PMC - PubMed
-
- Brown KRS, England KM, Goss KL, Snyder JM, Acarregui MJ. VEGF induces airway epithelial cell proliferation in human fetal lung in vitro. Am J Physiol Lung Cell Mol Physiol 281: L1001– L1010, 2001 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

