Decreased osteoclastogenesis in serotonin-deficient mice
- PMID: 22308416
- PMCID: PMC3289318
- DOI: 10.1073/pnas.1117792109
Decreased osteoclastogenesis in serotonin-deficient mice
Abstract
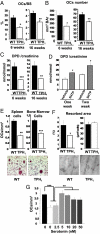
Peripheral serotonin, synthesized by tryptophan hydroxylase-1 (TPH(1)), has been shown to play a key role in several physiological functions. Recently, controversy has emerged about whether peripheral serotonin has any effect on bone density and remodeling.We therefore decided to investigate in detail bone remodeling in growing and mature TPH(1) knockout mice (TPH(1)(-/-)). Bone resorption in TPH(1)(-/-) mice, as assessed by biochemical markers and bone histomorphometry, was markedly decreased at both ages. Using bone marrow transplantation, we present evidence that the decrease in bone resorption in TPH(1)(-/-) mice is cell-autonomous. Cultures from TPH(1)(-/-) in the presence of macrophage colony-stimulating factor and receptor activator for NF-KB ligand (RANKL) displayed fewer osteoclasts, and the decreased differentiation could be rescued by adding serotonin. Our data also provide evidence that in the presence of RANKL, osteoclast precursors express TPH(1) and synthesize serotonin. Furthermore, pharmacological inhibition of serotonin receptor 1B with SB224289, and of receptor 2A with ketanserin, also reduced the number of osteoclasts. Our findings reveal that serotonin has an important local action in bone, as it can amplify the effect of RANKL on osteoclastogenesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


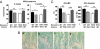
References
-
- Bianco P, Riminucci M, Gronthos S, Robey PG. Bone marrow stromal stem cells: Nature, biology, and potential applications. Stem Cells. 2001;19(3):180–192. - PubMed
-
- Teitelbaum SL. Bone resorption by osteoclasts. Science. 2000;289:1504–1508. - PubMed
-
- Khosla S. Minireview: The OPG/RANKL/RANK system. Endocrinology. 2001;142:5050–5055. - PubMed
-
- Kaumann AJ, Levy FO. 5-Hydroxytryptamine receptors in the human cardiovascular system. Pharmacol Ther. 2006;111:674–706. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

