The ancillary protein 1 of Streptococcus pyogenes FCT-1 pili mediates cell adhesion and biofilm formation through heterophilic as well as homophilic interactions
- PMID: 22320452
- PMCID: PMC3490378
- DOI: 10.1111/j.1365-2958.2012.07987.x
The ancillary protein 1 of Streptococcus pyogenes FCT-1 pili mediates cell adhesion and biofilm formation through heterophilic as well as homophilic interactions
Abstract
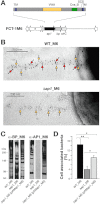
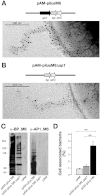
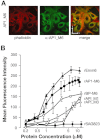
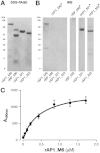
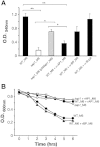
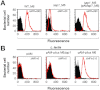
Gram-positive pili are known to play a role in bacterial adhesion to epithelial cells and in the formation of biofilm microbial communities. In the present study we undertook the functional characterization of the pilus ancillary protein 1 (AP1_M6) from Streptococcus pyogenes isolates expressing the FCT-1 pilus variant, known to be strong biofilm formers. Cell binding and biofilm formation assays using S. pyogenes in-frame deletion mutants, Lactococcus expressing heterologous FCT-1 pili and purified recombinant AP1_M6, indicated that this pilin is a strong cell adhesin that is also involved in bacterial biofilm formation. Moreover, we show that AP1_M6 establishes homophilic interactions that mediate inter-bacterial contact, possibly promoting bacterial colonization of target epithelial cells in the form of three-dimensional microcolonies. Finally, AP1_M6 knockout mutants were less virulent in mice, indicating that this protein is also implicated in GAS systemic infection.
© 2012 Blackwell Publishing Ltd.
Figures

Similar articles
-
Involvement of T6 pili in biofilm formation by serotype M6 Streptococcus pyogenes.J Bacteriol. 2012 Feb;194(4):804-12. doi: 10.1128/JB.06283-11. Epub 2011 Dec 9. J Bacteriol. 2012. PMID: 22155780 Free PMC article.
-
The Group A Streptococcus serotype M2 pilus plays a role in host cell adhesion and immune evasion.Mol Microbiol. 2017 Jan;103(2):282-298. doi: 10.1111/mmi.13556. Epub 2016 Nov 14. Mol Microbiol. 2017. PMID: 27741558
-
Streptococcus pyogenes pili promote pharyngeal cell adhesion and biofilm formation.Mol Microbiol. 2007 May;64(4):968-83. doi: 10.1111/j.1365-2958.2007.05704.x. Mol Microbiol. 2007. PMID: 17501921
-
Pili with strong attachments: Gram-positive bacteria do it differently.Mol Microbiol. 2006 Oct;62(2):320-30. doi: 10.1111/j.1365-2958.2006.05279.x. Epub 2006 Sep 15. Mol Microbiol. 2006. PMID: 16978260 Review.
-
Cellular interactions by LPxTG-anchored pneumococcal adhesins and their streptococcal homologues.Cell Microbiol. 2011 Feb;13(2):186-97. doi: 10.1111/j.1462-5822.2010.01560.x. Epub 2010 Dec 28. Cell Microbiol. 2011. PMID: 21199258 Review.
Cited by
-
Tissue tropisms in group A Streptococcus: what virulence factors distinguish pharyngitis from impetigo strains?Curr Opin Infect Dis. 2016 Jun;29(3):295-303. doi: 10.1097/QCO.0000000000000262. Curr Opin Infect Dis. 2016. PMID: 26895573 Free PMC article. Review.
-
Deciphering Streptococcal Biofilms.Microorganisms. 2020 Nov 21;8(11):1835. doi: 10.3390/microorganisms8111835. Microorganisms. 2020. PMID: 33233415 Free PMC article. Review.
-
Group A Streptococcus Pili-Roles in Pathogenesis and Potential for Vaccine Development.Microorganisms. 2024 Mar 11;12(3):555. doi: 10.3390/microorganisms12030555. Microorganisms. 2024. PMID: 38543606 Free PMC article. Review.
-
Galleria mellonella larvae as an infection model for group A streptococcus.Virulence. 2013 Jul 1;4(5):419-28. doi: 10.4161/viru.24930. Epub 2013 May 7. Virulence. 2013. PMID: 23652836 Free PMC article.
-
Novel Molecular Insights about Lactobacillar Sortase-Dependent Piliation.Int J Mol Sci. 2017 Jul 18;18(7):1551. doi: 10.3390/ijms18071551. Int J Mol Sci. 2017. PMID: 28718795 Free PMC article. Review.
References
-
- Abbot EL, Smith WD, Siou G, Chiriboga C, Smith RJ, Wilson JA, et al. Pili mediate specific adhesion of Streptococcus pyogenes to human tonsil and skin. Cell Microbiol. 2007;9:1822–1833. - PubMed
-
- Buccato S, Maione D, Rinaudo CD, Volpini G, Taddei AR, Rosini R, et al. Use of Lactococcus lactis expressing pili from group B Streptococcus as a broad-coverage vaccine against streptococcal disease. J Infect Dis. 2006;194:331–340. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

